Difference between revisions of "RocG"
(→Expression and regulation) |
|||
| Line 12: | Line 12: | ||
|style="background:#ABCDEF;" align="center"| '''Product''' || glutamate dehydrogenase (major) | |style="background:#ABCDEF;" align="center"| '''Product''' || glutamate dehydrogenase (major) | ||
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || arginine utilization, controls the activity of GltC | + | |style="background:#ABCDEF;" align="center"|'''Function''' || arginine utilization, controls the activity of [[GltC]] |
|- | |- | ||
|style="background:#ABCDEF;" align="center"| '''MW, pI''' || 46.2 kDa, 6.28 | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 46.2 kDa, 6.28 | ||
Revision as of 10:27, 12 March 2009
- Description: catabolic glutamate dehydrogenase induced by arginine, ornithine or proline, subject to carbon catabolite repression
| Gene name | rocG |
| Synonyms | |
| Essential | no |
| Product | glutamate dehydrogenase (major) |
| Function | arginine utilization, controls the activity of GltC |
| MW, pI | 46.2 kDa, 6.28 |
| Gene length, protein length | 1272 bp, 424 amino acids |
| Immediate neighbours | yweA, rocA |
| Gene sequence (+200bp) corrected | Protein sequence |
Genetic context 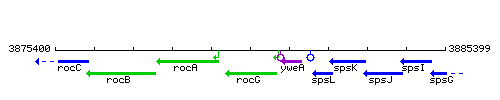
| |
Contents
The gene
Basic information
- Coordinates: 3879765 - 3881036
Phenotypes of a mutant
Poor growth on complex media such as SP (sporulation medium). No growth in minimal media with arginine as the only carbon source. Rapid accumulation of suppressor mutants (gudB1)
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: L-glutamate + H(2)O + NAD(+) = 2-oxoglutarate + NH(3) + NADH, controls the activity of the GltC transcription activator PubMed
- Protein family: Glu/Leu/Phe/Val dehydrogenases family
- Paralogous protein(s): GudB
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions: RocG-GltC, this interaction prevents transcription activation of the gltA-gltB operon by GltC PubMed
- Localization:
Database entries
- Structure:
- Swiss prot entry: [3]
- KEGG entry: [4]
- E.C. number: [5]
Additional information
Expression and regulation
- Operon: rocG
- Regulation: induced by arginine (RocR, AhrC), ornithine or proline, subject to carbon catabolite repression (CcpA)
- Regulatory mechanism: RocR: transcription activation PubMedPubMed; AhrC: transcription activation ; CcpA: transcription repression
- Additional information:
Activation by RocR requires binding of RocG to a downstream element PubMed
Biological materials
- Mutant: GP747 (spc), GP726 (aphA3), GP810 (tet) available in Stülke lab
- Expression vector: pGP902 (in pGP172, N-terminal Strep-tag), a series of rocG variants is also available in pGP172, available in Stülke lab
- lacZ fusion:
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- Antibody: available in Stülke lab
Labs working on this gene/protein
Linc Sonenshein, Tufts University, Boston, MA, USA Homepage
Jörg Stülke, University of Göttingen, Germany Homepage
Your additional remarks
References
Enzymatic activity of RocG
- Belitsky BR, Sonenshein AL (1998) Role and regulation of Bacillus subtilis glutamate dehydrogenase genes. J Bacteriol 180:6298-6305 PubMed
- Commichau, F. M., Gunka, K., Landmann, J. J. & Stülke, J. (2008) Glutamate metabolism in Bacillus subtilis: Gene expression and enzyme activities evolved to avoid futile cycles and to allow rapid responses to perturbations in the system. J. Bacteriol. 190: 3557-3564. PubMed
- Khan, M. I., K. Ito, H. Kim, H. Ashida, T. Ishikawa, H. Shibata, and Y. Sawa. 2005. Molecular properties and enhancement of thermostability by random mutagenesis of glutamate dehydrogenase from Bacillus subtilis. Biosci. Biotechnol. Biochem. 69: 1861-1870. PubMed
- Stillman TJ, Baker PJ, Britton KL, Rice DW Conformational flexibility in glutamate dehydrogenase. Role of water in substrate recognition and catalysis. J Mol Biol 1993, 234:1131-1139. PubMed
Function in the control of GltC activity
- Commichau, F. M., Wacker, I., Schleider, J., Blencke, H.-M., Reif, I., Tripal, P., and Stülke, J. (2007) Characterization of Bacillus subtilis mutants with carbon source-independent glutamate biosynthesis. J Mol Microbiol Biotechnol 12: 106-113. PubMed
- Commichau, F. M., Herzberg, C., Tripal, P., Valerius, O., and Stülke, J. (2007) A regulatory protein-protein interaction governs glutamate biosynthesis in Bacillus subtilis: The glutamate dehydrogenase RocG moonlights in controlling the transcription factor GltC. Mol Microbiol 65: 642-654. PubMed
- Herzberg, C., Flórez Weidinger, L. A., Dörrbecker, B., Hübner, S., Stülke, J. & Commichau, F. M. (2007) SPINE: A method for the rapid detection and analysis of protein-protein interactions in vivo. Proteomics 7: 4032-4035. PubMed
- Belitsky BR, Sonenshein AL (2004) Modulation of activity of Bacillus subtilis regulatory proteins GltC and TnrA by glutamate dehydrogenase. J Bacteriol 186:3399-3407 PubMed
Expression of rocG
- Ali, N. O., J. Jeusset, E. Larquet, E. le Cam, B. Belitsky, A. L. Sonenshein, T. Msadek, and M. Débarbouillé. 2003. Specificity of the interaction of RocR with the rocG-rocA intergenic region in Bacillus subtilis. Microbiology 149: 739-750. PubMed
- Belitsky BR, Sonenshein, AL: An enhancer element located downstream of the major glutamate dehydrogenase gene of Bacillus subtilis. Proc Natl Acad Sci USA 1999, 96:10290-10295. PubMed
- Belitsky BR, Sonenshein, AL: CcpA-dependent regulation of Bacillus subtilis glutamate dehydrogenase gene expression. J Bacteriol 2004, 186:3392-3398. PubMed