Difference between revisions of "PtkA"
(→Reviews) |
|||
| Line 45: | Line 45: | ||
= This gene is a member of the following [[regulons]] = | = This gene is a member of the following [[regulons]] = | ||
| − | {{SubtiWiki regulon|[[AbrB regulon]]}} | + | {{SubtiWiki regulon|[[AbrB regulon]]}}, |
| + | {{SubtiWiki regulon|[[DegU regulon]]}}, | ||
| + | {{SubtiWiki regulon|[[Spo0A regulon]]}} | ||
=The gene= | =The gene= | ||
| Line 116: | Line 118: | ||
* '''Regulatory mechanism:''' | * '''Regulatory mechanism:''' | ||
| − | ** [[DegU]]-P: transcription | + | ** [[Spo0A]]: transcription activation {{PubMed|26283769}} |
| + | ** [[DegU]]-P: transcription activation {{PubMed|26283769}} | ||
** [[AbrB]]: transcription repression {{PubMed|20817675}} | ** [[AbrB]]: transcription repression {{PubMed|20817675}} | ||
| Line 152: | Line 155: | ||
==Original publications== | ==Original publications== | ||
| − | <pubmed> 12970183, 15741737, 15866923, 17367396, 19258708 18547145 20497499 20509597 20815827 20817675 23939619 24493247 24728941 25278935 25374563 </pubmed> | + | <pubmed> 12970183, 15741737, 15866923, 17367396, 19258708 18547145 20497499 20509597 20815827 20817675 23939619 24493247 24728941 25278935 25374563 26283769</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 17:06, 19 August 2015
- Description: protein tyrosine kinase
| Gene name | ptkA |
| Synonyms | ywqD |
| Essential | no |
| Product | protein tyrosine kinase |
| Function | protein phosphorylation |
| Gene expression levels in SubtiExpress: ptkA | |
| Interactions involving this protein in SubtInteract: PtkA | |
| MW, pI | 25 kDa, 9.628 |
| Gene length, protein length | 711 bp, 237 aa |
| Immediate neighbours | ptpZ, tkmA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 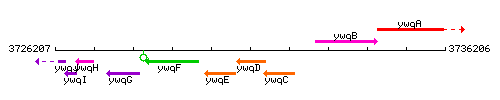
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
biofilm formation, protein modification, membrane proteins
This gene is a member of the following regulons
AbrB regulon, DegU regulon, Spo0A regulon
The gene
Basic information
- Locus tag: BSU36250
Phenotypes of a mutant
- Accumulation of extra chromosome equivalents PubMed
- Defect in biofilm formation, this involves the kinase activity, but the target protein is unknown PubMed
Database entries
- BsubCyc: BSU36250
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + a [protein]-L-tyrosine = ADP + a [protein]-L-tyrosine phosphate (according to Swiss-Prot), autophosphorylation, phosphorylation of Ugd, TuaD, Ssb, SsbB
- Protein family: BY-kinase, see the Bacterial Protein Tyrosine Kinase Database)
- Paralogous protein(s): EpsB
Extended information on the protein
- Kinetic information:
- Domains: single BY-kinase domain
- Modification: autophosphorylation at residues Y225, Y227 and Y228 (primary site) PubMed, dephosphorylated by PtpZ PubMed
- Cofactors: ATP
- Effectors of protein activity: TkmA - transmembrane modulator, activates PtkA autophosphorylation and substrate phosphorylation PubMed
Database entries
- BsubCyc: BSU36250
- Structure: 2VED (CapB, the homolog in Staphylococcus aureus)
- UniProt: P96716
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- KO strain created with pMUTIN-2, available from Ivan Mijakovic
- GP1520 (spc), available in Jörg Stülke's lab
- GP1544 (ermC), available in Jörg Stülke's lab
- GP1587 (cat) , available in Jörg Stülke's lab
- GP1521 epsB (aphA3) ptkA (spc) double mutant available in Jörg Stülke's lab
- GP1529 tkmA-ptkA::spc available in Jörg Stülke's lab
- GP1610 (ptkA-ptpZ, spc), available in Jörg Stülke's lab
- Expression vector: pQE-30, N-terminally 6xHis-tagged, available from Ivan Mijakovic
- lacZ fusion: in a KO strain created with pMUTIN-2, available from Ivan Mijakovic
- GFP fusion: CFP-fusion, available from Ivan Mijakovic
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Jörg Stülke's lab
- Antibody:
Labs working on this gene/protein
Ivan Mijakovic, Thiverval-Grignon, France
Your additional remarks
References
Reviews
Ivan Mijakovic, Josef Deutscher
Protein-tyrosine phosphorylation in Bacillus subtilis: a 10-year retrospective.
Front Microbiol: 2015, 6;18
[PubMed:25667587]
[WorldCat.org]
[DOI]
(P e)
Jan Gerwig, Jörg Stülke
Far from being well understood: multiple protein phosphorylation events control cell differentiation in Bacillus subtilis at different levels.
Front Microbiol: 2014, 5;704
[PubMed:25540643]
[WorldCat.org]
[DOI]
(P e)
Joseph D Chao, Dennis Wong, Yossef Av-Gay
Microbial protein-tyrosine kinases.
J Biol Chem: 2014, 289(14);9463-72
[PubMed:24554699]
[WorldCat.org]
[DOI]
(I p)
Jörg Stülke
More than just activity control: phosphorylation may control all aspects of a protein's properties.
Mol Microbiol: 2010, 77(2);273-5
[PubMed:20497498]
[WorldCat.org]
[DOI]
(I p)
Original publications
Tantan Gao, Jennifer Greenwich, Yan Li, Qi Wang, Yunrong Chai
The Bacterial Tyrosine Kinase Activator TkmA Contributes to Biofilm Formation Largely Independently of the Cognate Kinase PtkA in Bacillus subtilis.
J Bacteriol: 2015, 197(21);3421-32
[PubMed:26283769]
[WorldCat.org]
[DOI]
(I p)
Lei Shi, Nathalie Pigeonneau, Magali Ventroux, Abderahmane Derouiche, Vladimir Bidnenko, Ivan Mijakovic, Marie-Françoise Noirot-Gros
Protein-tyrosine phosphorylation interaction network in Bacillus subtilis reveals new substrates, kinase activators and kinase cross-talk.
Front Microbiol: 2014, 5;538
[PubMed:25374563]
[WorldCat.org]
[DOI]
(P e)
Lei Shi, Nathalie Pigeonneau, Vaishnavi Ravikumar, Paula Dobrinic, Boris Macek, Damjan Franjevic, Marie-Francoise Noirot-Gros, Ivan Mijakovic
Cross-phosphorylation of bacterial serine/threonine and tyrosine protein kinases on key regulatory residues.
Front Microbiol: 2014, 5;495
[PubMed:25278935]
[WorldCat.org]
[DOI]
(P e)
Lei Shi, Boyang Ji, Lorena Kolar-Znika, Ana Boskovic, Fanny Jadeau, Christophe Combet, Christophe Grangeasse, Damjan Franjevic, Emmanuel Talla, Ivan Mijakovic
Evolution of bacterial protein-tyrosine kinases and their relaxed specificity toward substrates.
Genome Biol Evol: 2014, 6(4);800-17
[PubMed:24728941]
[WorldCat.org]
[DOI]
(I p)
Jan Gerwig, Taryn B Kiley, Katrin Gunka, Nicola Stanley-Wall, Jörg Stülke
The protein tyrosine kinases EpsB and PtkA differentially affect biofilm formation in Bacillus subtilis.
Microbiology (Reading): 2014, 160(Pt 4);682-691
[PubMed:24493247]
[WorldCat.org]
[DOI]
(I p)
Abderahmane Derouiche, Vladimir Bidnenko, Rosa Grenha, Nathalie Pigonneau, Magali Ventroux, Mirita Franz-Wachtel, Sylvie Nessler, Marie-Françoise Noirot-Gros, Ivan Mijakovic
Interaction of bacterial fatty-acid-displaced regulators with DNA is interrupted by tyrosine phosphorylation in the helix-turn-helix domain.
Nucleic Acids Res: 2013, 41(20);9371-81
[PubMed:23939619]
[WorldCat.org]
[DOI]
(I p)
Onuma Chumsakul, Hiroki Takahashi, Taku Oshima, Takahiro Hishimoto, Shigehiko Kanaya, Naotake Ogasawara, Shu Ishikawa
Genome-wide binding profiles of the Bacillus subtilis transition state regulator AbrB and its homolog Abh reveals their interactive role in transcriptional regulation.
Nucleic Acids Res: 2011, 39(2);414-28
[PubMed:20817675]
[WorldCat.org]
[DOI]
(I p)
Taryn B Kiley, Nicola R Stanley-Wall
Post-translational control of Bacillus subtilis biofilm formation mediated by tyrosine phosphorylation.
Mol Microbiol: 2010, 78(4);947-63
[PubMed:20815827]
[WorldCat.org]
[DOI]
(I p)
Boumediene Soufi, Chanchal Kumar, Florian Gnad, Matthias Mann, Ivan Mijakovic, Boris Macek
Stable isotope labeling by amino acids in cell culture (SILAC) applied to quantitative proteomics of Bacillus subtilis.
J Proteome Res: 2010, 9(7);3638-46
[PubMed:20509597]
[WorldCat.org]
[DOI]
(I p)
Carsten Jers, Malene Mejer Pedersen, Dafni Katerina Paspaliari, Wolfgang Schütz, Christina Johnsson, Boumediene Soufi, Boris Macek, Peter Ruhdal Jensen, Ivan Mijakovic
Bacillus subtilis BY-kinase PtkA controls enzyme activity and localization of its protein substrates.
Mol Microbiol: 2010, 77(2);287-99
[PubMed:20497499]
[WorldCat.org]
[DOI]
(I p)
Dina Petranovic, Christophe Grangeasse, Boris Macek, Mohammad Abdillatef, Virginie Gueguen-Chaignon, Sylvie Nessler, Josef Deutscher, Ivan Mijakovic
Activation of Bacillus subtilis Ugd by the BY-kinase PtkA proceeds via phosphorylation of its residue tyrosine 70.
J Mol Microbiol Biotechnol: 2009, 17(2);83-9
[PubMed:19258708]
[WorldCat.org]
[DOI]
(I p)
Vanesa Olivares-Illana, Philippe Meyer, Emmanuelle Bechet, Virginie Gueguen-Chaignon, Didier Soulat, Sylvie Lazereg-Riquier, Ivan Mijakovic, Josef Deutscher, Alain J Cozzone, Olivier Laprévote, Solange Morera, Christophe Grangeasse, Sylvie Nessler
Structural basis for the regulation mechanism of the tyrosine kinase CapB from Staphylococcus aureus.
PLoS Biol: 2008, 6(6);e143
[PubMed:18547145]
[WorldCat.org]
[DOI]
(I p)
Dina Petranovic, Ole Michelsen, Ksenija Zahradka, Catarina Silva, Mirjana Petranovic, Peter Ruhdal Jensen, Ivan Mijakovic
Bacillus subtilis strain deficient for the protein-tyrosine kinase PtkA exhibits impaired DNA replication.
Mol Microbiol: 2007, 63(6);1797-805
[PubMed:17367396]
[WorldCat.org]
[DOI]
(P p)
Ivan Mijakovic, Lucia Musumeci, Lutz Tautz, Dina Petranovic, Robert A Edwards, Peter Ruhdal Jensen, Tomas Mustelin, Josef Deutscher, Nunzio Bottini
In vitro characterization of the Bacillus subtilis protein tyrosine phosphatase YwqE.
J Bacteriol: 2005, 187(10);3384-90
[PubMed:15866923]
[WorldCat.org]
[DOI]
(P p)
Ivan Mijakovic, Dina Petranovic, Josef Deutscher
How tyrosine phosphorylation affects the UDP-glucose dehydrogenase activity of Bacillus subtilis YwqF.
J Mol Microbiol Biotechnol: 2004, 8(1);19-25
[PubMed:15741737]
[WorldCat.org]
[DOI]
(P p)
Ivan Mijakovic, Sandrine Poncet, Grégory Boël, Alain Mazé, Sylvie Gillet, Emmanuel Jamet, Paulette Decottignies, Christophe Grangeasse, Patricia Doublet, Pierre Le Maréchal, Josef Deutscher
Transmembrane modulator-dependent bacterial tyrosine kinase activates UDP-glucose dehydrogenases.
EMBO J: 2003, 22(18);4709-18
[PubMed:12970183]
[WorldCat.org]
[DOI]
(P p)