Difference between revisions of "AccC"
(→References) |
(→Biological materials) |
||
| Line 128: | Line 128: | ||
=Biological materials = | =Biological materials = | ||
* '''Mutant:''' | * '''Mutant:''' | ||
| + | ** GP1478 (chromosomal ''accC''-Strep fusion, ''aphA''3), purification from ''B. subtilis'', for [[SPINE]], available in [[Jörg Stülke]]'s lab''' | ||
* '''Expression vector:''' | * '''Expression vector:''' | ||
| Line 135: | Line 136: | ||
* '''GFP fusion:''' | * '''GFP fusion:''' | ||
| − | * '''two-hybrid system:''' | + | * '''two-hybrid system:''' ''B. pertussis'' adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Jörg Stülke]]'s lab |
| + | |||
| + | * '''FLAG-tag construct:''' | ||
| + | ** GP1483 (''spc'', based on [[pGP1331]]), available in [[Jörg Stülke]]'s lab | ||
* '''Antibody:''' | * '''Antibody:''' | ||
Revision as of 10:20, 6 March 2015
- Description: acetyl-CoA carboxylase (biotin carboxylase subunit)
| Gene name | accC |
| Synonyms | yqhX |
| Essential | yes PubMed |
| Product | acetyl-CoA carboxylase (biotin carboxylase subunit) |
| Function | production of malonyl-CoA, the substrate for fatty acid biosynthesis |
| Gene expression levels in SubtiExpress: accC | |
| Interactions involving this protein in SubtInteract: AccC | |
| Metabolic function and regulation of this protein in SubtiPathways: accC | |
| MW, pI | 49 kDa, 4.72 |
| Gene length, protein length | 1350 bp, 450 aa |
| Immediate neighbours | yqhY, accB |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 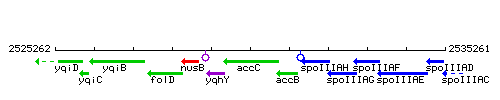
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed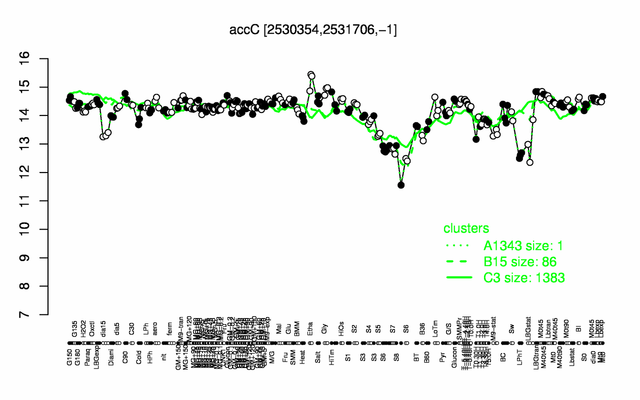
| |
Contents
Categories containing this gene/protein
biosynthesis of lipids, essential genes
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU24340
Phenotypes of a mutant
essential PubMed
Database entries
- BsubCyc: BSU24340
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + biotin-carboxyl-carrier protein + CO2 = ADP + phosphate + carboxybiotin-carboxyl-carrier protein (according to Swiss-Prot)
- Protein family:
- Paralogous protein(s): YotE
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- BsubCyc: BSU24340
- UniProt: P49787
- KEGG entry: [3]
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 527 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 286 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 634 PubMed
Biological materials
- Mutant:
- GP1478 (chromosomal accC-Strep fusion, aphA3), purification from B. subtilis, for SPINE, available in Jörg Stülke's lab
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Jörg Stülke's lab
- FLAG-tag construct:
- GP1483 (spc, based on pGP1331), available in Jörg Stülke's lab
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
L Tong
Acetyl-coenzyme A carboxylase: crucial metabolic enzyme and attractive target for drug discovery.
Cell Mol Life Sci: 2005, 62(16);1784-803
[PubMed:15968460]
[WorldCat.org]
[DOI]
(P p)
Stephen W White, Jie Zheng, Yong-Mei Zhang, Rock
The structural biology of type II fatty acid biosynthesis.
Annu Rev Biochem: 2005, 74;791-831
[PubMed:15952903]
[WorldCat.org]
[DOI]
(P p)
John E Cronan, Grover L Waldrop
Multi-subunit acetyl-CoA carboxylases.
Prog Lipid Res: 2002, 41(5);407-35
[PubMed:12121720]
[WorldCat.org]
[DOI]
(P p)
Original Publications
P Marini, S J Li, D Gardiol, J E Cronan, D de Mendoza
The genes encoding the biotin carboxyl carrier protein and biotin carboxylase subunits of Bacillus subtilis acetyl coenzyme A carboxylase, the first enzyme of fatty acid synthesis.
J Bacteriol: 1995, 177(23);7003-6
[PubMed:7592499]
[WorldCat.org]
[DOI]
(P p)
G L Waldrop, I Rayment, H M Holden
Three-dimensional structure of the biotin carboxylase subunit of acetyl-CoA carboxylase.
Biochemistry: 1994, 33(34);10249-56
[PubMed:7915138]
[WorldCat.org]
[DOI]
(P p)