Difference between revisions of "EpsE"
(→Reviews) |
|||
| Line 46: | Line 46: | ||
{{SubtiWiki category|[[motility and chemotaxis]]}}, | {{SubtiWiki category|[[motility and chemotaxis]]}}, | ||
{{SubtiWiki category|[[biofilm formation]]}}, | {{SubtiWiki category|[[biofilm formation]]}}, | ||
| − | {{SubtiWiki category|[[membrane proteins]]}} | + | {{SubtiWiki category|[[membrane proteins]]}}, |
| + | {{SubtiWiki category|[[phosphoproteins]]}} | ||
= This gene is a member of the following [[regulons]] = | = This gene is a member of the following [[regulons]] = | ||
| Line 91: | Line 92: | ||
* '''Domains:''' | * '''Domains:''' | ||
| − | * '''Modification:''' | + | * '''Modification:''' phosphorylated by [[EpsB]] on a Tyr residue {{PubMed|25085422}} |
* '''Cofactor(s):''' | * '''Cofactor(s):''' | ||
| Line 164: | Line 165: | ||
<pubmed>20374491 20230605 </pubmed> | <pubmed>20374491 20230605 </pubmed> | ||
===Other original publications=== | ===Other original publications=== | ||
| − | <pubmed>22113911 15661000,16430695,18047568, 18566286 18647168 21170308 20817675 21856853 21815947 23646920</pubmed> | + | <pubmed>22113911 15661000,16430695,18047568, 18566286 18647168 21170308 20817675 21856853 21815947 23646920 25085422</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 10:30, 5 August 2014
- Description: inhibitor of motility and glycosyltransferase required for EPS biosynthesis
| Gene name | epsE |
| Synonyms | yveO |
| Essential | no |
| Product | glycosyltransferase, inhibitor of motility |
| Function | biofilm formation |
| Gene expression levels in SubtiExpress: epsE | |
| Interactions involving this protein in SubtInteract: EpsE | |
| Regulation of this protein in SubtiPathways: epsE | |
| MW, pI | 32 kDa, 9.804 |
| Gene length, protein length | 834 bp, 278 aa |
| Immediate neighbours | epsF, epsD |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 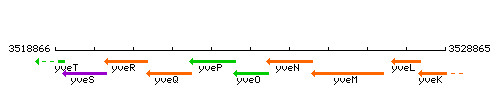
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
motility and chemotaxis, biofilm formation, membrane proteins, phosphoproteins
This gene is a member of the following regulons
AbrB regulon, EAR riboswitch, RemA regulon, SinR regulon
The gene
Basic information
- Locus tag: BSU34330
Phenotypes of a mutant
- smooth colonies on MsGG medium, no biofilm formation PubMed
Database entries
- BsubCyc: BSU34330
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: glycosyltransferase 2 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cell membrane, forms spots at flagellar basal bodies PubMed
Database entries
- BsubCyc: BSU34330
- Structure:
- UniProt: P71054
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulatory mechanism:
- Additional information:
- induction by sequestration of SinR by SinI or SlrA PubMed
- the epsA-epsB-epsC-epsD-epsE-epsF-epsG-epsH-epsI-epsJ-epsK-epsL-epsM-epsN-epsO operon is not expressed in a ymdB mutant PubMed
- the amount of the mRNA is substantially decreased upon depletion of RNase Y (this is likely due to the increased stability of the sinR mRNA) PubMed
- the EAR riboswitch (eps-associated RNA switch) located between epsB and epsC mediates processive antitermination and allows expression of the long eps operon PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
- Daniel Kearns, Indiana University, Bloomington, USA, homepage
- Richard Losick, Harvard Univ., Cambridge, USA homepage
Your additional remarks
References
Reviews
Original publications
The EAR RNA switch
Other original publications