Difference between revisions of "LysA"
| Line 121: | Line 121: | ||
** number of protein molecules per cell (minimal medium with glucose and ammonium): 627 {{PubMed|24696501}} | ** number of protein molecules per cell (minimal medium with glucose and ammonium): 627 {{PubMed|24696501}} | ||
** number of protein molecules per cell (complex medium with amino acids, without glucose): 2286 {{PubMed|24696501}} | ** number of protein molecules per cell (complex medium with amino acids, without glucose): 2286 {{PubMed|24696501}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 5990 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 1660 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 5360 {{PubMed|21395229}} | ||
=Biological materials = | =Biological materials = | ||
| − | |||
* '''Mutant:''' | * '''Mutant:''' | ||
** 1A615 ( ''lysA''::''erm''), {{PubMed|3015878}}, available at [http://pasture.asc.ohio-state.edu/BGSC/getdetail.cfm?bgscid=1A615&Search=1A615 BGSC] | ** 1A615 ( ''lysA''::''erm''), {{PubMed|3015878}}, available at [http://pasture.asc.ohio-state.edu/BGSC/getdetail.cfm?bgscid=1A615&Search=1A615 BGSC] | ||
Revision as of 14:09, 17 April 2014
- Description: diaminopimelate decarboxylase
| Gene name | lysA |
| Synonyms | |
| Essential | no |
| Product | diaminopimelate decarboxylase |
| Function | biosynthesis of lysine |
| Gene expression levels in SubtiExpress: lysA | |
| Metabolic function and regulation of this protein in SubtiPathways: lysA | |
| MW, pI | 48 kDa, 5.007 |
| Gene length, protein length | 1317 bp, 439 aa |
| Immediate neighbours | ypuA, spoVAF |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 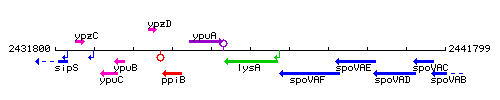
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed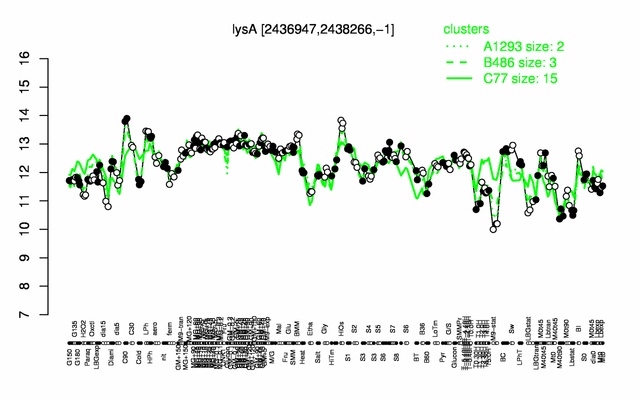
| |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids, sporulation proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU23380
Phenotypes of a mutant
- auxotrophic for lysine PubMed
Database entries
- BsubCyc: BSU23380
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Meso-2,6-diaminoheptanedioate = L-lysine + CO2 (according to Swiss-Prot)
- Protein family: Orn/Lys/Arg decarboxylase class-II family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Modification:
- Effectors of protein activity:
Database entries
- BsubCyc: BSU23380
- UniProt: P23630
- KEGG entry: [3]
- E.C. number: 4.1.1.20
Additional information
Expression and regulation
- Additional information:
- number of protein molecules per cell (minimal medium with glucose and ammonium): 627 PubMed
- number of protein molecules per cell (complex medium with amino acids, without glucose): 2286 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 5990 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 1660 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 5360 PubMed
Biological materials
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References