Difference between revisions of "DhbE"
| Line 12: | Line 12: | ||
|style="background:#ABCDEF;" align="center"| '''Product''' || 2,3-dihydroxybenzoate-AMP ligase (enterobactin synthetase component E) | |style="background:#ABCDEF;" align="center"| '''Product''' || 2,3-dihydroxybenzoate-AMP ligase (enterobactin synthetase component E) | ||
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || siderophore | + | |style="background:#ABCDEF;" align="center"|'''Function''' || biosynthesis of the siderophore bacillibactin |
|- | |- | ||
|style="background:#ABCDEF;" align="center"| '''MW, pI''' || 59 kDa, 5.684 | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 59 kDa, 5.684 | ||
Revision as of 09:29, 23 January 2010
- Description: 2,3-dihydroxybenzoate-AMP ligase (enterobactin synthetase component E)
| Gene name | dhbE |
| Synonyms | entE |
| Essential | no |
| Product | 2,3-dihydroxybenzoate-AMP ligase (enterobactin synthetase component E) |
| Function | biosynthesis of the siderophore bacillibactin |
| MW, pI | 59 kDa, 5.684 |
| Gene length, protein length | 1617 bp, 539 aa |
| Immediate neighbours | dhbB, dhbC |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 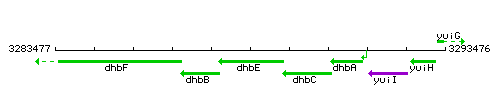
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU31980
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: ATP-dependent AMP-binding enzyme family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization: cytoplasm (according to Swiss-Prot)
Database entries
- UniProt: P40871
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Mohamed Marahiel, Marburg University, Germany homepage
Your additional remarks
References