Difference between revisions of "LonA"
(→Additional information) |
|||
| Line 92: | Line 92: | ||
=Expression and regulation= | =Expression and regulation= | ||
| + | * '''Operon:''' ''[[lonA]]-[[ysxC]]'' {{PubMed|7961402}} | ||
| − | + | * '''[[Sigma factor]]:''' [[SigA]] {{PubMed|7961402}} | |
| − | |||
| − | * '''[[Sigma factor]]:''' | ||
* '''Regulation:''' | * '''Regulation:''' | ||
| + | ** induced by heat stress ([[CtsR]]) {{PubMed|9852015}} | ||
* '''Regulatory mechanism:''' | * '''Regulatory mechanism:''' | ||
| + | ** [[CtsR]]: transcription repression {{PubMed|9852015}} | ||
* '''Additional information:''' | * '''Additional information:''' | ||
| Line 123: | Line 124: | ||
=References= | =References= | ||
| − | <pubmed>7961403,7961402, 19542270, 19643080 </pubmed> | + | <pubmed>7961403,7961402, 19542270, 19643080 9852015</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 20:07, 13 December 2009
- Description: class III heat-shock ATP-dependent protease
| Gene name | lonA |
| Synonyms | lon |
| Essential | no |
| Product | class III heat-shock ATP-dependent protease |
| Function | protein quality control |
| Regulation of this protein in SubtiPathways: Stress | |
| MW, pI | 86 kDa, 5.774 |
| Gene length, protein length | 2322 bp, 774 aa |
| Immediate neighbours | ysxC, lonB |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 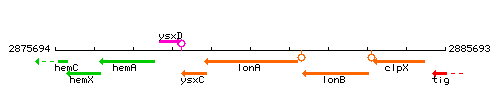
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU28200
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
A mutation in lonA suppresses the motility defect of a lytC mutant PubMed
Evidence for a specific DNA binding activitity of the α-domain was found PubMed
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Hydrolysis of proteins in presence of ATP (according to Swiss-Prot)
- Protein family: peptidase S16 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization:
Database entries
- Structure: 1X37 (Ssd domain)
- UniProt: P37945
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References