Difference between revisions of "Sandbox"
(Replaced content with "Basura") |
|||
| Line 1: | Line 1: | ||
| − | + | * '''Description:''' enolase, glycolytic/ gluconeogenic enzyme, [[universally conserved protein]]<br/><br/> | |
| + | |||
| + | {| align="right" border="1" cellpadding="2" | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"|'''Gene name''' | ||
| + | |''eno'' | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' '' | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Essential''' || no | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Product''' || enolase | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"|'''Function''' || enzyme in glycolysis/ gluconeogenesis | ||
| + | |- | ||
| + | |colspan="2" style="background:#FAF8CC;" align="center"| '''Interactions involving this protein in [http://cellpublisher.gobics.de/subtinteract/startpage/start/ ''Subt''Interact]''': [http://cellpublisher.gobics.de/subtinteract/interactionList/2/Eno Eno] | ||
| + | |- | ||
| + | |colspan="2" style="background:#FAF8CC;" align="center"| '''Metabolic function and regulation of this protein in [[SubtiPathways|''Subti''Pathways]]: <br/>[http://subtiwiki.uni-goettingen.de/pathways/carbon_flow.html Central C-metabolism]''' | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 46,4 kDa, 4.49 | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 1290 bp, 430 amino acids | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[yvbK]]'', ''[[pgm]]'' | ||
| + | |- | ||
| + | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB15395]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' | ||
| + | |- | ||
| + | |colspan="2" | '''Genetic context''' <br/> [[Image:eno_context.gif]] | ||
| + | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
| + | |} | ||
| + | |||
| + | <br/><br/><br/><br/> | ||
| + | |||
| + | __TOC__ | ||
| + | |||
| + | |||
| + | |||
| + | <br/><br/><br/><br/> | ||
| + | |||
| + | |||
| + | = [[Categories]] containing this gene/protein = | ||
| + | {{SubtiWiki category|[[carbon core metabolism]]}}, | ||
| + | {{SubtiWiki category|[[membrane proteins]]}}, | ||
| + | {{SubtiWiki category|[[phosphoproteins]]}}, | ||
| + | {{SubtiWiki category|[[universally conserved proteins]]}} | ||
| + | |||
| + | = This gene is a member of the following [[regulons]] = | ||
| + | {{SubtiWiki regulon|[[CggR regulon]]}} | ||
| + | |||
| + | =The gene= | ||
| + | |||
| + | === Basic information === | ||
| + | * '''Locus tag:''' BSU33900 | ||
| + | |||
| + | ===Phenotypes of a mutant === | ||
| + | * no growth on LB, requires glucose and malate | ||
| + | * essential according to Kobayashi et al. on LB [http://www.ncbi.nlm.nih.gov/pubmed/12682299 PubMed] | ||
| + | |||
| + | === Database entries === | ||
| + | |||
| + | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/cggR-gapA-pgk-tpiA-pgm-eno.html] | ||
| + | |||
| + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10899] | ||
| + | |||
| + | === Additional information=== | ||
| + | |||
| + | =The protein= | ||
| + | |||
| + | === Basic information/ Evolution === | ||
| + | |||
| + | * '''Catalyzed reaction/ biological activity:''' 2-phospho-D-glycerate = phosphoenolpyruvate + H<sub>2</sub>O (according to Swiss-Prot) 2-phospho-D-glycerate = phosphoenolpyruvate + H(2)O | ||
| + | |||
| + | * '''Protein family:''' enolase family (according to Swiss-Prot) | ||
| + | |||
| + | * '''Paralogous protein(s):''' | ||
| + | |||
| + | === Extended information on the protein === | ||
| + | |||
| + | * '''Kinetic information:''' reversible Michaelis-Menten [http://www.ncbi.nlm.nih.gov/sites/entrez/25885 PubMed] | ||
| + | |||
| + | * '''Domains:''' | ||
| + | ** substrate binding domain (366–369) | ||
| + | |||
| + | * '''Modification:''' phosphorylation on Thr-141 AND Ser-259 AND Tyr-281 AND Ser-325 [http://www.ncbi.nlm.nih.gov/sites/entrez/17218307 PubMed], [http://www.ncbi.nlm.nih.gov/pubmed/16493705 PubMed], [http://www.ncbi.nlm.nih.gov/pubmed/17726680 PubMed] | ||
| + | |||
| + | * '''Cofactor(s):''' Mg2+ | ||
| + | |||
| + | * '''Effectors of protein activity:''' | ||
| + | ** Inhibited by EDTA [http://www.ncbi.nlm.nih.gov/sites/entrez/25885 PubMed] | ||
| + | |||
| + | * '''[[SubtInteract|Interactions]]:''' | ||
| + | ** forms octamers {{PubMed|22198292}} | ||
| + | ** part of the [[RNA degradosome]] {{PubMed|19193632}} | ||
| + | ** [[Eno]]-[[PfkA]] {{PubMed|19193632}}, K(D) of the interaction: 40 nM {{PubMed|22198292}} | ||
| + | ** [[Eno]]-[[Rny]] {{PubMed|19193632,21803996}}, K(D) of the interaction: 100 nM {{PubMed|22198292}} | ||
| + | ** [[Eno]]-[[CshA]] {{PubMed|20572937}} | ||
| + | |||
| + | * '''[[Localization]]:''' | ||
| + | ** cytoplasm [http://www.ncbi.nlm.nih.gov/sites/entrez/16479537 PubMed] | ||
| + | ** membrane associated [http://www.ncbi.nlm.nih.gov/sites/entrez/18763711 PubMed] | ||
| + | ** exported, this requires a long, unbent α-helix (from A108 to L126) {{PubMed|15003462,21856851}} | ||
| + | |||
| + | === Database entries === | ||
| + | |||
| + | * '''Structure:''' | ||
| + | ** [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=4A3R 4A3R] {{PubMed|22198292}} | ||
| + | ** [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=1W6T 1W6T] (from ''Streptococcus pneumoniae'') {{PubMed|15476816}} | ||
| + | |||
| + | * '''UniProt:''' [http://www.uniprot.org/uniprot/P37869 P37869] | ||
| + | |||
| + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu:BSU33900] | ||
| + | |||
| + | * '''E.C. number:''' [http://www.expasy.org/enzyme/4.2.1.11 4.2.1.11] | ||
| + | |||
| + | === Additional information=== | ||
| + | * Enolase is a [[moonlighting proteins|moonlighting protein]]. [http://www.ncbi.nlm.nih.gov/sites/entrez/19193632 PubMed] | ||
| + | * There are indications that this enzyme is an octamer [http://www.ncbi.nlm.nih.gov/sites/entrez/25885 PubMed] | ||
| + | * [[universally conserved protein]] | ||
| + | * extensive information on the structure and enzymatic properties of Eno can be found at [http://www.proteopedia.org/wiki/index.php/Enolase Proteopedia] | ||
| + | |||
| + | =Expression and regulation= | ||
| + | |||
| + | * '''Operon:''' | ||
| + | ** ''[[cggR]]-[[gapA]]-[[pgk]]-[[tpiA]]-[[pgm]]-[[eno]]'' {{PubMed|11489127}} | ||
| + | ** ''[[pgk]]-[[tpiA]]-[[pgm]]-[[eno]]'' {{PubMed|11489127}} | ||
| + | |||
| + | * '''Sigma factor:''' [[SigA]] {{PubMed|11489127}} | ||
| + | |||
| + | * '''Regulation:''' | ||
| + | ** expression activated by glucose (3.3 fold) [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed] | ||
| + | ** ''[[cggR]]'': induced by glycolytic substrates [[CggR]] {{PubMed|11489127}} | ||
| + | ** ''[[pgk]]'': constitutive {{PubMed|11489127}} | ||
| + | |||
| + | * '''Regulatory mechanism:''' transcription repression by [[CggR]] {{PubMed|11489127}} | ||
| + | |||
| + | * '''Additional information:''' | ||
| + | |||
| + | =Biological materials = | ||
| + | |||
| + | * '''Mutant:''' | ||
| + | ** GP594 (''eno''::''cat''), available in [[Stülke]] lab | ||
| + | ** GP599 (''eno''::''erm''), available in [[Stülke]] lab | ||
| + | ** GP698 (''eno''-''[[pgm]]''::''cat''), available in [[Stülke]] lab | ||
| + | |||
| + | * '''Expression vector:''' | ||
| + | ** pGP1426 (expression of ''[[eno]]'' in ''B. subtilis'', in [[pBQ200]]), available in [[Stülke]] lab | ||
| + | ** pGP1500 (expression of ''[[pgm]]'' and ''[[eno]]'' in ''B. subtilis'', in [[pBQ200]]), available in [[Stülke]] lab | ||
| + | ** pGP563 (N-terminal His-tag, in [[pWH844]]), available in [[Stülke]] lab | ||
| + | ** pGP1276 (N-terminal Strep-tag, purification from ''E. coli'', in [[pGP172]]), available in [[Stülke]] lab | ||
| + | ** pGP93 (N-terminal Strep-tag, purification from ''B. subtilis'', for [[SPINE]], in [[pGP380]]), available in [[Stülke]] lab | ||
| + | ** GP1215 (''eno''-''Strep'' ''(spc)''), purification from ''B. subtilis'', for [[SPINE]], available in [[Stülke]] lab | ||
| + | |||
| + | * '''lacZ fusion:''' | ||
| + | ** see ''[[pgk]]'' | ||
| + | |||
| + | * '''GFP fusion:''' pHT315-yfp-eno, available in [[Mijakovic]] lab | ||
| + | |||
| + | * '''two-hybrid system:''' ''B. pertussis'' adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Stülke]] lab | ||
| + | |||
| + | * '''FLAG-tag construct:''' GP1214 (spc, based on [[pGP1331]]), available in the [[Stülke]] lab | ||
| + | |||
| + | * '''Antibody:''' available in [[Stülke]] lab | ||
| + | |||
| + | =Labs working on this gene/protein= | ||
| + | |||
| + | [[Stülke|Jörg Stülke]], University of Göttingen, Germany | ||
| + | [http://wwwuser.gwdg.de/~genmibio/stuelke.html Homepage] | ||
| + | |||
| + | =Your additional remarks= | ||
| + | |||
| + | =References= | ||
| + | ==Reviews== | ||
| + | <pubmed> 8994873 </pubmed> | ||
| + | ==Subcellular localization of enolase== | ||
| + | '''Additional publications:''' {{PubMed|21856851}} | ||
| + | <pubmed> 16479537 18763711 15003462 20497499 </pubmed> | ||
| + | |||
| + | ==Other original publications== | ||
| + | <pubmed> 17726680, 17218307, 12850135, 19193632, 11489127, 8021172, 17505547, 25885, 20572937 15476816 9988532 ,21803996 22198292 </pubmed> | ||
| + | |||
| + | [[Category:Protein-coding genes]] | ||
Revision as of 15:46, 22 March 2012
- Description: enolase, glycolytic/ gluconeogenic enzyme, universally conserved protein
| Gene name | eno |
| Synonyms | |
| Essential | no |
| Product | enolase |
| Function | enzyme in glycolysis/ gluconeogenesis |
| Interactions involving this protein in SubtInteract: Eno | |
| Metabolic function and regulation of this protein in SubtiPathways: Central C-metabolism | |
| MW, pI | 46,4 kDa, 4.49 |
| Gene length, protein length | 1290 bp, 430 amino acids |
| Immediate neighbours | yvbK, pgm |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 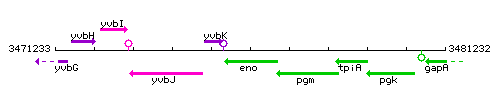
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
carbon core metabolism, membrane proteins, phosphoproteins, universally conserved proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU33900
Phenotypes of a mutant
- no growth on LB, requires glucose and malate
- essential according to Kobayashi et al. on LB PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 2-phospho-D-glycerate = phosphoenolpyruvate + H2O (according to Swiss-Prot) 2-phospho-D-glycerate = phosphoenolpyruvate + H(2)O
- Protein family: enolase family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information: reversible Michaelis-Menten PubMed
- Domains:
- substrate binding domain (366–369)
- Cofactor(s): Mg2+
- Effectors of protein activity:
- Inhibited by EDTA PubMed
Database entries
- UniProt: P37869
- KEGG entry: [3]
- E.C. number: 4.2.1.11
Additional information
- Enolase is a moonlighting protein. PubMed
- There are indications that this enzyme is an octamer PubMed
- universally conserved protein
- extensive information on the structure and enzymatic properties of Eno can be found at Proteopedia
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- pGP1426 (expression of eno in B. subtilis, in pBQ200), available in Stülke lab
- pGP1500 (expression of pgm and eno in B. subtilis, in pBQ200), available in Stülke lab
- pGP563 (N-terminal His-tag, in pWH844), available in Stülke lab
- pGP1276 (N-terminal Strep-tag, purification from E. coli, in pGP172), available in Stülke lab
- pGP93 (N-terminal Strep-tag, purification from B. subtilis, for SPINE, in pGP380), available in Stülke lab
- GP1215 (eno-Strep (spc)), purification from B. subtilis, for SPINE, available in Stülke lab
- lacZ fusion:
- see pgk
- GFP fusion: pHT315-yfp-eno, available in Mijakovic lab
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- Antibody: available in Stülke lab
Labs working on this gene/protein
Jörg Stülke, University of Göttingen, Germany Homepage
Your additional remarks
References
Reviews
Subcellular localization of enolase
Additional publications: PubMed
Other original publications
Joseph A Newman, Lorraine Hewitt, Cecilia Rodrigues, Alexandra S Solovyova, Colin R Harwood, Richard J Lewis
Dissection of the network of interactions that links RNA processing with glycolysis in the Bacillus subtilis degradosome.
J Mol Biol: 2012, 416(1);121-36
[PubMed:22198292]
[WorldCat.org]
[DOI]
(I p)
Martin Lehnik-Habrink, Joseph Newman, Fabian M Rothe, Alexandra S Solovyova, Cecilia Rodrigues, Christina Herzberg, Fabian M Commichau, Richard J Lewis, Jörg Stülke
RNase Y in Bacillus subtilis: a Natively disordered protein that is the functional equivalent of RNase E from Escherichia coli.
J Bacteriol: 2011, 193(19);5431-41
[PubMed:21803996]
[WorldCat.org]
[DOI]
(I p)
Martin Lehnik-Habrink, Henrike Pförtner, Leonie Rempeters, Nico Pietack, Christina Herzberg, Jörg Stülke
The RNA degradosome in Bacillus subtilis: identification of CshA as the major RNA helicase in the multiprotein complex.
Mol Microbiol: 2010, 77(4);958-71
[PubMed:20572937]
[WorldCat.org]
[DOI]
(I p)
Fabian M Commichau, Fabian M Rothe, Christina Herzberg, Eva Wagner, Daniel Hellwig, Martin Lehnik-Habrink, Elke Hammer, Uwe Völker, Jörg Stülke
Novel activities of glycolytic enzymes in Bacillus subtilis: interactions with essential proteins involved in mRNA processing.
Mol Cell Proteomics: 2009, 8(6);1350-60
[PubMed:19193632]
[WorldCat.org]
[DOI]
(I p)
Christine Eymann, Dörte Becher, Jörg Bernhardt, Katrin Gronau, Anja Klutzny, Michael Hecker
Dynamics of protein phosphorylation on Ser/Thr/Tyr in Bacillus subtilis.
Proteomics: 2007, 7(19);3509-26
[PubMed:17726680]
[WorldCat.org]
[DOI]
(P p)
Laurent Jannière, Danielle Canceill, Catherine Suski, Sophie Kanga, Bérengère Dalmais, Roxane Lestini, Anne-Françoise Monnier, Jérôme Chapuis, Alexander Bolotin, Marina Titok, Emmanuelle Le Chatelier, S Dusko Ehrlich
Genetic evidence for a link between glycolysis and DNA replication.
PLoS One: 2007, 2(5);e447
[PubMed:17505547]
[WorldCat.org]
[DOI]
(I e)
Boris Macek, Ivan Mijakovic, Jesper V Olsen, Florian Gnad, Chanchal Kumar, Peter R Jensen, Matthias Mann
The serine/threonine/tyrosine phosphoproteome of the model bacterium Bacillus subtilis.
Mol Cell Proteomics: 2007, 6(4);697-707
[PubMed:17218307]
[WorldCat.org]
[DOI]
(P p)
Stefanie Ehinger, Wolf-Dieter Schubert, Simone Bergmann, Sven Hammerschmidt, Dirk W Heinz
Plasmin(ogen)-binding alpha-enolase from Streptococcus pneumoniae: crystal structure and evaluation of plasmin(ogen)-binding sites.
J Mol Biol: 2004, 343(4);997-1005
[PubMed:15476816]
[WorldCat.org]
[DOI]
(P p)
Hans-Matti Blencke, Georg Homuth, Holger Ludwig, Ulrike Mäder, Michael Hecker, Jörg Stülke
Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways.
Metab Eng: 2003, 5(2);133-49
[PubMed:12850135]
[WorldCat.org]
[DOI]
(P p)
H Ludwig, G Homuth, M Schmalisch, F M Dyka, M Hecker, J Stülke
Transcription of glycolytic genes and operons in Bacillus subtilis: evidence for the presence of multiple levels of control of the gapA operon.
Mol Microbiol: 2001, 41(2);409-22
[PubMed:11489127]
[WorldCat.org]
[DOI]
(P p)
C K Brown, P L Kuhlman, S Mattingly, K Slates, P J Calie, W W Farrar
A model of the quaternary structure of enolases, based on structural and evolutionary analysis of the octameric enolase from Bacillus subtilis.
J Protein Chem: 1998, 17(8);855-66
[PubMed:9988532]
[WorldCat.org]
[DOI]
(P p)
M A Leyva-Vazquez, P Setlow
Cloning and nucleotide sequences of the genes encoding triose phosphate isomerase, phosphoglycerate mutase, and enolase from Bacillus subtilis.
J Bacteriol: 1994, 176(13);3903-10
[PubMed:8021172]
[WorldCat.org]
[DOI]
(P p)
R P Singh, P Setlow
Enolase from spores and cells of Bacillus megaterium: two-step purification of the enzyme and some of its properties.
J Bacteriol: 1978, 134(1);353-5
[PubMed:25885]
[WorldCat.org]
[DOI]
(P p)