Difference between revisions of "ClpX"
(→Original Publications) |
|||
| Line 156: | Line 156: | ||
<pubmed>23375660 19680248 17302811 23479438,19609260</pubmed> | <pubmed>23375660 19680248 17302811 23479438,19609260</pubmed> | ||
==Original Publications== | ==Original Publications== | ||
| − | <pubmed>12761164 10809708,9643546,11807061,14679237,18689476,16899079,8973311, 19136590 , 11325926 8973311 9852015 18689473 20525796 15948963 18786145 24417481 24942655 </pubmed> | + | <pubmed>12761164 10809708,9643546,11807061,14679237,18689476,16899079,8973311, 19136590 , 11325926 8973311 9852015 18689473 20525796 15948963 18786145 24417481 24942655 25433860 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 18:17, 5 December 2014
- Description: ATP-dependent Clp protease ATP-binding subunit (class III heat-shock protein)
| Gene name | clpX |
| Synonyms | |
| Essential | no |
| Product | ATP-dependent Clp protease ATP-binding subunit |
| Function | protein degradation |
| Gene expression levels in SubtiExpress: clpX | |
| Interactions involving this protein in SubtInteract: ClpX | |
| Metabolic function and regulation of this protein in SubtiPathways: clpX | |
| MW, pI | 46 kDa, 4.645 |
| Gene length, protein length | 1260 bp, 420 aa |
| Immediate neighbours | lonB, tig |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 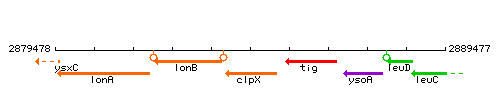
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed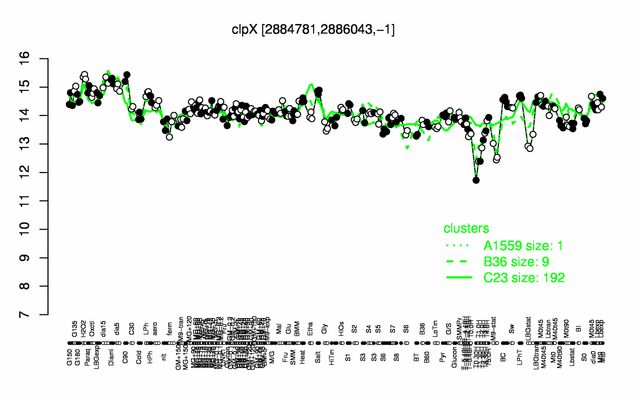
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU28220
Phenotypes of a mutant
- increased thermotolerance due to increased stabiliy of Spx and thus increased expression of trxA PubMed
Database entries
- BsubCyc: BSU28220
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATPase/chaperone
- Protein family: clpX chaperone family (according to Swiss-Prot) ClpX (IP004487) InterPro, AAA+ -type ATPase (IPR013093) InterPro (PF07724) PFAM
Targets of ClpX-ClpP-dependent protein degradation
Extended information on the protein
- Kinetic information:
- Modification:
- Effectors of protein activity:
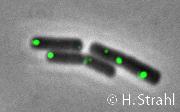
- Localization: cytoplasmic polar clusters, excluded from the nucleoid, induced clustering upon heat shock, colocalization with ClpP PubMed
Database entries
- BsubCyc: BSU28220
- Structure: homologue structure resolved 1UM8, structural model of B. subtilis ClpX available from hstrahl
- UniProt: P50866
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: clpX PubMed
- Additional information:
Biological materials
- Mutant: clpX::kan, clpX::spec and clpX::cat available from the Hamoen] Lab
- Expression vector:
- lacZ fusion:
- GFP fusion: C-terminal GFP fusions (both single copy and 2th copy in amyE locus, also as CFP and YFP variants) available from the Hamoen] Lab
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Leendert Hamoen, Newcastle University, UK homepage
Your additional remarks
References
Reviews
Original Publications
Yuh Shiwa, Hirofumi Yoshikawa, Teruo Tanaka, Mitsuo Ogura
Bacillus subtilis degSU operon is regulated by the ClpXP-Spx regulated proteolysis system.
J Biochem: 2015, 157(5);321-30
[PubMed:25433860]
[WorldCat.org]
[DOI]
(I p)
Chio Mui Chan, Erik Hahn, Peter Zuber
Adaptor bypass mutations of Bacillus subtilis spx suggest a mechanism for YjbH-enhanced proteolysis of the regulator Spx by ClpXP.
Mol Microbiol: 2014, 93(3);426-38
[PubMed:24942655]
[WorldCat.org]
[DOI]
(I p)
Stephanie Runde, Noël Molière, Anja Heinz, Etienne Maisonneuve, Armgard Janczikowski, Alexander K W Elsholz, Ulf Gerth, Michael Hecker, Kürşad Turgay
The role of thiol oxidative stress response in heat-induced protein aggregate formation during thermotolerance in Bacillus subtilis.
Mol Microbiol: 2014, 91(5);1036-52
[PubMed:24417481]
[WorldCat.org]
[DOI]
(I p)
Irnov Irnov, Cynthia M Sharma, Jörg Vogel, Wade C Winkler
Identification of regulatory RNAs in Bacillus subtilis.
Nucleic Acids Res: 2010, 38(19);6637-51
[PubMed:20525796]
[WorldCat.org]
[DOI]
(I p)
Daniel P Haeusser, Amy H Lee, Richard B Weart, Petra Anne Levin
ClpX inhibits FtsZ assembly in a manner that does not require its ATP hydrolysis-dependent chaperone activity.
J Bacteriol: 2009, 191(6);1986-91
[PubMed:19136590]
[WorldCat.org]
[DOI]
(I p)
Janine Kirstein, Henrik Strahl, Noël Molière, Leendert W Hamoen, Kürşad Turgay
Localization of general and regulatory proteolysis in Bacillus subtilis cells.
Mol Microbiol: 2008, 70(3);682-94
[PubMed:18786145]
[WorldCat.org]
[DOI]
(I p)
James Kain, Gina G He, Richard Losick
Polar localization and compartmentalization of ClpP proteases during growth and sporulation in Bacillus subtilis.
J Bacteriol: 2008, 190(20);6749-57
[PubMed:18689476]
[WorldCat.org]
[DOI]
(I p)
Lyle A Simmons, Alan D Grossman, Graham C Walker
Clp and Lon proteases occupy distinct subcellular positions in Bacillus subtilis.
J Bacteriol: 2008, 190(20);6758-68
[PubMed:18689473]
[WorldCat.org]
[DOI]
(I p)
Stephan Zellmeier, Wolfgang Schumann, Thomas Wiegert
Involvement of Clp protease activity in modulating the Bacillus subtilissigmaw stress response.
Mol Microbiol: 2006, 61(6);1569-82
[PubMed:16899079]
[WorldCat.org]
[DOI]
(P p)
Richard B Weart, Shunji Nakano, Brooke E Lane, Peter Zuber, Petra Anne Levin
The ClpX chaperone modulates assembly of the tubulin-like protein FtsZ.
Mol Microbiol: 2005, 57(1);238-49
[PubMed:15948963]
[WorldCat.org]
[DOI]
(P p)
Ulf Gerth, Janine Kirstein, Jörg Mostertz, Torsten Waldminghaus, Marcus Miethke, Holger Kock, Michael Hecker
Fine-tuning in regulation of Clp protein content in Bacillus subtilis.
J Bacteriol: 2004, 186(1);179-91
[PubMed:14679237]
[WorldCat.org]
[DOI]
(P p)
Hideaki Nanamiya, Emiko Shiomi, Mitsuo Ogura, Teruo Tanaka, Kei Asai, Fujio Kawamura
Involvement of ClpX protein in the post-transcriptional regulation of a competence specific transcription factor, ComK protein, of Bacillus subtilis.
J Biochem: 2003, 133(3);295-302
[PubMed:12761164]
[WorldCat.org]
[DOI]
(P p)
Tiina Pummi, Soile Leskelä, Eva Wahlström, Ulf Gerth, Harold Tjalsma, Michael Hecker, Matti Sarvas, Vesa P Kontinen
ClpXP protease regulates the signal peptide cleavage of secretory preproteins in Bacillus subtilis with a mechanism distinct from that of the Ecs ABC transporter.
J Bacteriol: 2002, 184(4);1010-8
[PubMed:11807061]
[WorldCat.org]
[DOI]
(P p)
M Serrano, S Hövel, C P Moran, A O Henriques, U Völker
Forespore-specific transcription of the lonB gene during sporulation in Bacillus subtilis.
J Bacteriol: 2001, 183(10);2995-3003
[PubMed:11325926]
[WorldCat.org]
[DOI]
(P p)
E Krüger, E Witt, S Ohlmeier, R Hanschke, M Hecker
The clp proteases of Bacillus subtilis are directly involved in degradation of misfolded proteins.
J Bacteriol: 2000, 182(11);3259-65
[PubMed:10809708]
[WorldCat.org]
[DOI]
(P p)
E Krüger, M Hecker
The first gene of the Bacillus subtilis clpC operon, ctsR, encodes a negative regulator of its own operon and other class III heat shock genes.
J Bacteriol: 1998, 180(24);6681-8
[PubMed:9852015]
[WorldCat.org]
[DOI]
(P p)
U Gerth, E Krüger, I Derré, T Msadek, M Hecker
Stress induction of the Bacillus subtilis clpP gene encoding a homologue of the proteolytic component of the Clp protease and the involvement of ClpP and ClpX in stress tolerance.
Mol Microbiol: 1998, 28(4);787-802
[PubMed:9643546]
[WorldCat.org]
[DOI]
(P p)
U Gerth, A Wipat, C R Harwood, N Carter, P T Emmerson, M Hecker
Sequence and transcriptional analysis of clpX, a class-III heat-shock gene of Bacillus subtilis.
Gene: 1996, 181(1-2);77-83
[PubMed:8973311]
[WorldCat.org]
[DOI]
(P p)