Difference between revisions of "CtsR"
| Line 61: | Line 61: | ||
=== Database entries === | === Database entries === | ||
| + | * '''BsubCyc:''' [http://bsubcyc.org/BSUB/NEW-IMAGE?type=NIL&object=BSU00830&redirect=T BSU00830] | ||
* '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ctsR-mcsAB-clpC-radA-yacK.html] | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ctsR-mcsAB-clpC-radA-yacK.html] | ||
| Line 103: | Line 104: | ||
=== Database entries === | === Database entries === | ||
| + | * '''BsubCyc:''' [http://bsubcyc.org/BSUB/NEW-IMAGE?type=NIL&object=BSU00830&redirect=T BSU00830] | ||
* '''Structure:''' [http://www.pdb.org/pdb/explore/explore.do?structureId=3H0D 3H0D] (complex with a 26bp DNA duplex, from ''Geobacillus stearothermophilus'') {{PubMed|19498169}} | * '''Structure:''' [http://www.pdb.org/pdb/explore/explore.do?structureId=3H0D 3H0D] (complex with a 26bp DNA duplex, from ''Geobacillus stearothermophilus'') {{PubMed|19498169}} | ||
Revision as of 12:47, 2 April 2014
| Gene name | ctsR |
| Synonyms | yacG |
| Essential | no |
| Product | transcription repressor |
| Function | regulation of protein degradation |
| Gene expression levels in SubtiExpress: ctsR | |
| Interactions involving this protein in SubtInteract: CtsR | |
| Regulatory function and regulation of this protein in SubtiPathways: ctsR | |
| MW, pI | 17 kDa, 9.261 |
| Gene length, protein length | 462 bp, 154 aa |
| Immediate neighbours | rrnW-5S, mcsA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 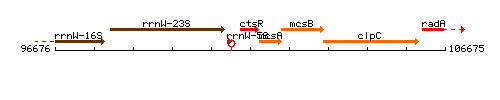
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed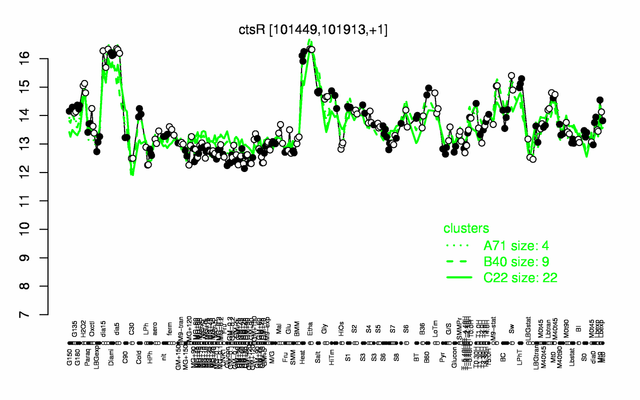
| |
Contents
Categories containing this gene/protein
proteolysis, transcription factors and their control, general stress proteins (controlled by SigB), heat shock proteins, phosphoproteins
This gene is a member of the following regulons
The CtsR regulon
The gene
Basic information
- Locus tag: BSU00830
Phenotypes of a mutant
Database entries
- BsubCyc: BSU00830
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: ctsR family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- BsubCyc: BSU00830
- UniProt: P37568
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information: the mRNA is very stable (half-life > 15 min) PubMed
Biological materials
- Mutant: ctsR::aphA3 availbale from the Gerth lab
ctsRG65P::spec available from the Gerth lab
- Expression vector: for expression, purification in E. coli with N-terminal His-tag, pRSETA available in Gerth lab
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody: available in Gerth lab
Labs working on this gene/protein
Your additional remarks
References
Reviews
Additional reviews: PubMed
Aurelia Battesti, Susan Gottesman
Roles of adaptor proteins in regulation of bacterial proteolysis.
Curr Opin Microbiol: 2013, 16(2);140-7
[PubMed:23375660]
[WorldCat.org]
[DOI]
(I p)
Alexander K W Elsholz, Ulf Gerth, Michael Hecker
Regulation of CtsR activity in low GC, Gram+ bacteria.
Adv Microb Physiol: 2010, 57;119-44
[PubMed:21078442]
[WorldCat.org]
[DOI]
(I p)
Wolfgang Schumann
The Bacillus subtilis heat shock stimulon.
Cell Stress Chaperones: 2003, 8(3);207-17
[PubMed:14984053]
[WorldCat.org]
[DOI]
(P p)
Original publications
Additional publications: PubMed