Difference between revisions of "Easter egg"
| Line 1: | Line 1: | ||
| − | + | * '''Description:''' enolase, glycolytic/ gluconeogenic enzyme, [[universally conserved protein]]<br/><br/> | |
| + | |||
| + | {| align="right" border="1" cellpadding="2" | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"|'''Gene name''' | ||
| + | |''eno'' | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' '' | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Essential''' || no | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Product''' || enolase | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"|'''Function''' || enzyme in glycolysis/ gluconeogenesis | ||
| + | |- | ||
| + | |colspan="2" style="background:#FAF8CC;" align="center"| '''Metabolic function and regulation of this protein in [[SubtiPathways|''Subti''Pathways]]: <br/>[http://subtiwiki.uni-goettingen.de/pathways/carbon_flow.html Central C-metabolism]''' | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 46,4 kDa, 4.49 | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 1290 bp, 430 amino acids | ||
| + | |- | ||
| + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[yvbK]]'', ''[[pgm]]'' | ||
| + | |- | ||
| + | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB15395]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' | ||
| + | |- | ||
| + | |colspan="2" | '''Genetic context''' <br/> [[Image:eno_context.gif]] | ||
| + | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
| + | |- | ||
| + | |} | ||
| + | |||
| + | __TOC__ | ||
| + | |||
| + | <br/><br/><br/><br/><br/><br/> | ||
| + | |||
| + | |||
| + | =The gene= | ||
| + | |||
| + | === Basic information === | ||
| + | |||
| + | * '''Locus tag:''' BSU33900 | ||
| + | |||
| + | ===Phenotypes of a mutant === | ||
| + | * no growth on LB, requires glucose and malate | ||
| + | * essential according to Kobayashi et al. on LB [http://www.ncbi.nlm.nih.gov/pubmed/12682299 PubMed] | ||
| + | |||
| + | === Database entries === | ||
| + | |||
| + | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/cggR-gapA-pgk-tpiA-pgm-eno.html] | ||
| + | |||
| + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10899] | ||
| + | |||
| + | === Additional information=== | ||
| + | |||
| + | =The protein= | ||
| + | |||
| + | === Basic information/ Evolution === | ||
| + | |||
| + | * '''Catalyzed reaction/ biological activity:''' 2-phospho-D-glycerate = phosphoenolpyruvate + H<sub>2</sub>O (according to Swiss-Prot) 2-phospho-D-glycerate = phosphoenolpyruvate + H(2)O | ||
| + | |||
| + | * '''Protein family:''' enolase family (according to Swiss-Prot) | ||
| + | |||
| + | * '''Paralogous protein(s):''' | ||
| + | |||
| + | === Extended information on the protein === | ||
| + | |||
| + | * '''Kinetic information:''' reversible Michaelis-Menten [http://www.ncbi.nlm.nih.gov/sites/entrez/25885 PubMed] | ||
| + | |||
| + | * '''Domains:''' | ||
| + | ** substrate binding domain (366–369) | ||
| + | |||
| + | * '''Modification:''' phosphorylation on Thr-141 AND Ser-259 AND Tyr-281 AND Ser-325 [http://www.ncbi.nlm.nih.gov/sites/entrez/17218307 PubMed], [http://www.ncbi.nlm.nih.gov/pubmed/16493705 PubMed], [http://www.ncbi.nlm.nih.gov/pubmed/17726680 PubMed] | ||
| + | |||
| + | * '''Cofactor(s):''' Mg2+ | ||
| + | |||
| + | * '''Effectors of protein activity:''' | ||
| + | ** Inhibited by EDTA [http://www.ncbi.nlm.nih.gov/sites/entrez/25885 PubMed] | ||
| + | |||
| + | * '''Interactions:''' [[Eno]]-[[PfkA]] {{PubMed|19193632}}, [[Eno]]-[[Rny]] {{PubMed|19193632}}, [[Eno]]-[[CshA]] {{PubMed|20572937}} | ||
| + | |||
| + | * '''Localization:''' cytoplasm [http://www.ncbi.nlm.nih.gov/sites/entrez/16479537 PubMed] and membrane associated [http://www.ncbi.nlm.nih.gov/sites/entrez/18763711 PubMed], exported {{PubMed|15003462}} | ||
| + | |||
| + | === Database entries === | ||
| + | |||
| + | * '''Structure:''' [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=1W6T 1W6T] (from ''Streptococcus pneumoniae'') {{PubMed|15476816}} | ||
| + | |||
| + | * '''UniProt:''' [http://www.uniprot.org/uniprot/P37869 P37869] | ||
| + | |||
| + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu:BSU33900] | ||
| + | |||
| + | * '''E.C. number:''' [http://www.expasy.org/enzyme/4.2.1.11 4.2.1.11] | ||
| + | |||
| + | === Additional information=== | ||
| + | * Enolase is a [[moonlighting proteins|moonlighting protein]]. [http://www.ncbi.nlm.nih.gov/sites/entrez/19193632 PubMed] | ||
| + | * There are indications that this enzyme is an octamer [http://www.ncbi.nlm.nih.gov/sites/entrez/25885 PubMed] | ||
| + | * [[universally conserved protein]] | ||
| + | |||
| + | =Expression and regulation= | ||
| + | |||
| + | * '''Operon:''' | ||
| + | ** ''[[cggR]]-[[gapA]]-[[pgk]]-[[tpiA]]-[[pgm]]-[[eno]]'' {{PubMed|11489127}} | ||
| + | ** ''[[pgk]]-[[tpiA]]-[[pgm]]-[[eno]]'' {{PubMed|11489127}} | ||
| + | |||
| + | * '''Sigma factor:''' [[SigA]] {{PubMed|11489127}} | ||
| + | |||
| + | * '''Regulation:''' | ||
| + | ** expression activated by glucose (3.3 fold) [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed] | ||
| + | ** ''[[cggR]]'': induced by glycolytic substrates [[CggR]] {{PubMed|11489127}} | ||
| + | ** ''[[pgk]]'': constitutive {{PubMed|11489127}} | ||
| + | |||
| + | * '''Regulatory mechanism:''' transcription repression by [[CggR]] {{PubMed|11489127}} | ||
| + | |||
| + | * '''Additional information:''' | ||
| + | |||
| + | =Biological materials = | ||
| + | |||
| + | * '''Mutant:''' | ||
| + | ** GP594 (''eno''::''cat''), available in [[Stülke]] lab | ||
| + | ** GP599 (''eno''::''erm''), available in [[Stülke]] lab | ||
| + | ** GP698 (''eno''-''[[pgm]]''::''cat''), available in [[Stülke]] lab | ||
| + | |||
| + | * '''Expression vector:''' | ||
| + | ** pGP1426 (expression of ''eno'' in ''B. subtilis'', in [[pBQ200]]), available in [[Stülke]] lab | ||
| + | ** pGP399 (expression of ''eno'' from ''E. coli'' in ''B. subtilis'', in [[pBQ200]]), available in [[Stülke]] lab | ||
| + | ** pGP563 (N-terminal His-tag, in [[pWH844]]), available in [[Stülke]] lab | ||
| + | ** pGP93 (N-terminal Strep-tag, purification from ''B. subtilis'', for [[SPINE]], in [[pGP380]]), available in [[Stülke]] lab | ||
| + | ** pGP1500 (expression of ''[[pgm]]'' and ''[[eno]]'' in ''B. subtilis'', in [[pBQ200]]), available in [[Stülke]] lab | ||
| + | |||
| + | * '''lacZ fusion:''' | ||
| + | ** see ''[[pgk]]'' | ||
| + | |||
| + | * '''GFP fusion:''' pHT315-yfp-eno, available in [[Mijakovic]] lab | ||
| + | |||
| + | * '''two-hybrid system:''' ''B. pertussis'' adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Stülke]] lab | ||
| + | |||
| + | * '''Antibody:''' available in [[Stülke]] lab | ||
| + | |||
| + | =Labs working on this gene/protein= | ||
| + | |||
| + | [[Stülke|Jörg Stülke]], University of Göttingen, Germany | ||
| + | [http://wwwuser.gwdg.de/~genmibio/stuelke.html Homepage] | ||
| + | |||
| + | =Your additional remarks= | ||
| + | |||
| + | =References= | ||
| + | ==Reviews== | ||
| + | <pubmed> 8994873 </pubmed> | ||
| + | ==Subcellular localization of enolase== | ||
| + | <pubmed> 16479537 18763711 15003462 20497499 </pubmed> | ||
| + | ==Other original publications== | ||
| + | <pubmed> 17726680, 17218307, 12850135, 19193632, 11489127, 8021172, 17505547, 25885, 20572937 15476816 9988532 </pubmed> | ||
| + | |||
| + | [[Category:Protein-coding genes]] | ||
Revision as of 13:12, 29 November 2010
- Description: enolase, glycolytic/ gluconeogenic enzyme, universally conserved protein
| Gene name | eno |
| Synonyms | |
| Essential | no |
| Product | enolase |
| Function | enzyme in glycolysis/ gluconeogenesis |
| Metabolic function and regulation of this protein in SubtiPathways: Central C-metabolism | |
| MW, pI | 46,4 kDa, 4.49 |
| Gene length, protein length | 1290 bp, 430 amino acids |
| Immediate neighbours | yvbK, pgm |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 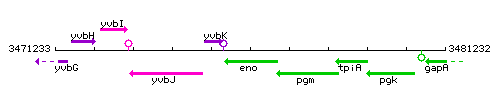
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU33900
Phenotypes of a mutant
- no growth on LB, requires glucose and malate
- essential according to Kobayashi et al. on LB PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 2-phospho-D-glycerate = phosphoenolpyruvate + H2O (according to Swiss-Prot) 2-phospho-D-glycerate = phosphoenolpyruvate + H(2)O
- Protein family: enolase family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information: reversible Michaelis-Menten PubMed
- Domains:
- substrate binding domain (366–369)
- Cofactor(s): Mg2+
- Effectors of protein activity:
- Inhibited by EDTA PubMed
Database entries
- UniProt: P37869
- KEGG entry: [3]
- E.C. number: 4.2.1.11
Additional information
- Enolase is a moonlighting protein. PubMed
- There are indications that this enzyme is an octamer PubMed
- universally conserved protein
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- pGP1426 (expression of eno in B. subtilis, in pBQ200), available in Stülke lab
- pGP399 (expression of eno from E. coli in B. subtilis, in pBQ200), available in Stülke lab
- pGP563 (N-terminal His-tag, in pWH844), available in Stülke lab
- pGP93 (N-terminal Strep-tag, purification from B. subtilis, for SPINE, in pGP380), available in Stülke lab
- pGP1500 (expression of pgm and eno in B. subtilis, in pBQ200), available in Stülke lab
- lacZ fusion:
- see pgk
- GFP fusion: pHT315-yfp-eno, available in Mijakovic lab
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- Antibody: available in Stülke lab
Labs working on this gene/protein
Jörg Stülke, University of Göttingen, Germany Homepage
Your additional remarks
References
Reviews
G H Reed, R R Poyner, T M Larsen, J E Wedekind, I Rayment
Structural and mechanistic studies of enolase.
Curr Opin Struct Biol: 1996, 6(6);736-43
[PubMed:8994873]
[WorldCat.org]
[DOI]
(P p)
Subcellular localization of enolase
Other original publications