KinA
- Description: two-component sensor kinase, phosphorylates Spo0F, part of the phosphorelay
| Gene name | kinA |
| Synonyms | spoIIF, spoIIJ, gsiC, scoB, scoD |
| Essential | no |
| Product | two-component sensor kinase |
| Function | initiation of sporulation |
| Gene expression levels in SubtiExpress: kinA | |
| Interactions involving this protein in SubtInteract: KinA | |
| Function and regulation of this protein in SubtiPathways: kinA | |
| MW, pI | 68 kDa, 5.491 |
| Gene length, protein length | 1818 bp, 606 aa |
| Immediate neighbours | pbpH, patA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 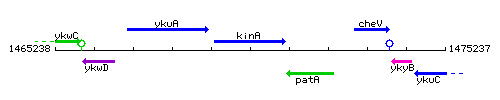
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed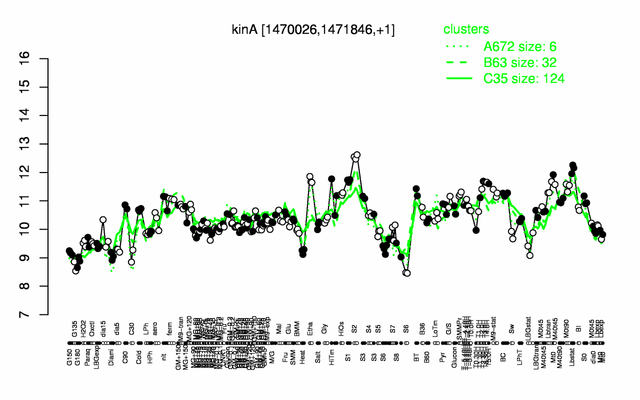
| |
Contents
Categories containing this gene/protein
protein modification, transcription factors and their control, phosphorelay, phosphoproteins
This gene is a member of the following regulons
SigH regulon, Spo0A regulon, stringent response
The gene
Basic information
- Locus tag: BSU13990
Phenotypes of a mutant
Database entries
- BsubCyc: BSU13990
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- three tandem PAS domains in the N-terminal region of KinA, the second PAS domain is the major N-terminal determinant of KinA dimerization PubMed
- the first PAS domain is required for NAD(+) binding PubMed
- C-terminal histidine phosphotransferase domain
- Modification: autophosphorylation on a His residue
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- cytoplasm
Database entries
- BsubCyc: BSU13990
- Structure: 2VLG (PAS domain)
- UniProt: P16497
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- expressed under conditions that trigger sporulation (Spo0A) PubMed
- induced upon addition of decoyinine (positive stringent response) PubMed
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original Publications
Seram Nganbiton Devi, Brittany Kiehler, Lindsey Haggett, Masaya Fujita
Evidence that Autophosphorylation of the Major Sporulation Kinase in Bacillus subtilis Is Able To Occur in trans.
J Bacteriol: 2015, 197(16);2675-84
[PubMed:26055117]
[WorldCat.org]
[DOI]
(I p)
Kristina M Boguslawski, Patrick A Hill, Kevin L Griffith
Novel mechanisms of controlling the activities of the transcription factors Spo0A and ComA by the plasmid-encoded quorum sensing regulators Rap60-Phr60 in Bacillus subtilis.
Mol Microbiol: 2015, 96(2);325-48
[PubMed:25598361]
[WorldCat.org]
[DOI]
(I p)
Ilana Kolodkin-Gal, Alexander K W Elsholz, Christine Muth, Peter R Girguis, Roberto Kolter, Richard Losick
Respiration control of multicellularity in Bacillus subtilis by a complex of the cytochrome chain with a membrane-embedded histidine kinase.
Genes Dev: 2013, 27(8);887-99
[PubMed:23599347]
[WorldCat.org]
[DOI]
(I p)
Brit Winnen, Eric Anderson, James L Cole, Glenn F King, Susan L Rowland
Role of the PAS sensor domains in the Bacillus subtilis sporulation kinase KinA.
J Bacteriol: 2013, 195(10);2349-58
[PubMed:23504013]
[WorldCat.org]
[DOI]
(I p)
Shigeo Tojo, Kazutake Hirooka, Yasutaro Fujita
Expression of kinA and kinB of Bacillus subtilis, necessary for sporulation initiation, is under positive stringent transcription control.
J Bacteriol: 2013, 195(8);1656-65
[PubMed:23378509]
[WorldCat.org]
[DOI]
(I p)
Sharon Garti-Levi, Ashlee Eswara, Yoav Smith, Masaya Fujita, Sigal Ben-Yehuda
Novel modulators controlling entry into sporulation in Bacillus subtilis.
J Bacteriol: 2013, 195(7);1475-83
[PubMed:23335417]
[WorldCat.org]
[DOI]
(I p)
Jatin Narula, Seram N Devi, Masaya Fujita, Oleg A Igoshin
Ultrasensitivity of the Bacillus subtilis sporulation decision.
Proc Natl Acad Sci U S A: 2012, 109(50);E3513-22
[PubMed:23169620]
[WorldCat.org]
[DOI]
(I p)
Angel E Dago, Alexander Schug, Andrea Procaccini, James A Hoch, Martin Weigt, Hendrik Szurmant
Structural basis of histidine kinase autophosphorylation deduced by integrating genomics, molecular dynamics, and mutagenesis.
Proc Natl Acad Sci U S A: 2012, 109(26);E1733-42
[PubMed:22670053]
[WorldCat.org]
[DOI]
(I p)
Joe H Levine, Michelle E Fontes, Jonathan Dworkin, Michael B Elowitz
Pulsed feedback defers cellular differentiation.
PLoS Biol: 2012, 10(1);e1001252
[PubMed:22303282]
[WorldCat.org]
[DOI]
(I p)
Prahathees Eswaramoorthy, Ashlee Dravis, Seram Nganbiton Devi, Monika Vishnoi, Hoang-Anh Dao, Masaya Fujita
Expression level of a chimeric kinase governs entry into sporulation in Bacillus subtilis.
J Bacteriol: 2011, 193(22);6113-22
[PubMed:21926229]
[WorldCat.org]
[DOI]
(I p)
Anna L McLoon, Ilana Kolodkin-Gal, Shmuel M Rubinstein, Roberto Kolter, Richard Losick
Spatial regulation of histidine kinases governing biofilm formation in Bacillus subtilis.
J Bacteriol: 2011, 193(3);679-85
[PubMed:21097618]
[WorldCat.org]
[DOI]
(I p)
Prahathees Eswaramoorthy, Daniel Duan, Jeffrey Dinh, Ashlee Dravis, Seram Nganbiton Devi, Masaya Fujita
The threshold level of the sensor histidine kinase KinA governs entry into sporulation in Bacillus subtilis.
J Bacteriol: 2010, 192(15);3870-82
[PubMed:20511506]
[WorldCat.org]
[DOI]
(I p)
Prahathees Eswaramoorthy, Jeffrey Dinh, Daniel Duan, Oleg A Igoshin, Masaya Fujita
Single-cell measurement of the levels and distributions of the phosphorelay components in a population of sporulating Bacillus subtilis cells.
Microbiology (Reading): 2010, 156(Pt 8);2294-2304
[PubMed:20413551]
[WorldCat.org]
[DOI]
(I p)
Prahathees Eswaramoorthy, Masaya Fujita
Systematic domain deletion analysis of the major sporulation kinase in Bacillus subtilis.
J Bacteriol: 2010, 192(6);1744-8
[PubMed:20081035]
[WorldCat.org]
[DOI]
(I p)
Prahathees Eswaramoorthy, Tao Guo, Masaya Fujita
In vivo domain-based functional analysis of the major sporulation sensor kinase, KinA, in Bacillus subtilis.
J Bacteriol: 2009, 191(17);5358-68
[PubMed:19561131]
[WorldCat.org]
[DOI]
(I p)
Kazuo Kobayashi, Ritsuko Kuwana, Hiromu Takamatsu
kinA mRNA is missing a stop codon in the undomesticated Bacillus subtilis strain ATCC 6051.
Microbiology (Reading): 2008, 154(Pt 1);54-63
[PubMed:18174125]
[WorldCat.org]
[DOI]
(P p)
Andrew E Whitten, David A Jacques, Boualem Hammouda, Tracey Hanley, Glenn F King, J Mitchell Guss, Jill Trewhella, David B Langley
The structure of the KinA-Sda complex suggests an allosteric mechanism of histidine kinase inhibition.
J Mol Biol: 2007, 368(2);407-20
[PubMed:17350039]
[WorldCat.org]
[DOI]
(P p)
Masaya Fujita, Richard Losick
Evidence that entry into sporulation in Bacillus subtilis is governed by a gradual increase in the level and activity of the master regulator Spo0A.
Genes Dev: 2005, 19(18);2236-44
[PubMed:16166384]
[WorldCat.org]
[DOI]
(P p)
Susan L Rowland, William F Burkholder, Katherine A Cunningham, Mark W Maciejewski, Alan D Grossman, Glenn F King
Structure and mechanism of action of Sda, an inhibitor of the histidine kinases that regulate initiation of sporulation in Bacillus subtilis.
Mol Cell: 2004, 13(5);689-701
[PubMed:15023339]
[WorldCat.org]
[DOI]
(P p)
Sophie J Stephenson, Marta Perego
Interaction surface of the Spo0A response regulator with the Spo0E phosphatase.
Mol Microbiol: 2002, 44(6);1455-67
[PubMed:12067336]
[WorldCat.org]
[DOI]
(P p)
K Stephenson, J A Hoch
PAS-A domain of phosphorelay sensor kinase A: a catalytic ATP-binding domain involved in the initiation of development in Bacillus subtilis.
Proc Natl Acad Sci U S A: 2001, 98(26);15251-6
[PubMed:11734624]
[WorldCat.org]
[DOI]
(P p)
L Wang, C Fabret, K Kanamaru, K Stephenson, V Dartois, M Perego, J A Hoch
Dissection of the functional and structural domains of phosphorelay histidine kinase A of Bacillus subtilis.
J Bacteriol: 2001, 183(9);2795-802
[PubMed:11292798]
[WorldCat.org]
[DOI]
(P p)
M Jiang, W Shao, M Perego, J A Hoch
Multiple histidine kinases regulate entry into stationary phase and sporulation in Bacillus subtilis.
Mol Microbiol: 2000, 38(3);535-42
[PubMed:11069677]
[WorldCat.org]
[DOI]
(P p)
C Fabret, V A Feher, J A Hoch
Two-component signal transduction in Bacillus subtilis: how one organism sees its world.
J Bacteriol: 1999, 181(7);1975-83
[PubMed:10094672]
[WorldCat.org]
[DOI]
(P p)
M Fujita, Y Sadaie
Feedback loops involving Spo0A and AbrB in in vitro transcription of the genes involved in the initiation of sporulation in Bacillus subtilis.
J Biochem: 1998, 124(1);98-104
[PubMed:9644251]
[WorldCat.org]
[DOI]
(P p)
C E Grimshaw, S Huang, C G Hanstein, M A Strauch, D Burbulys, L Wang, J A Hoch, J M Whiteley
Synergistic kinetic interactions between components of the phosphorelay controlling sporulation in Bacillus subtilis.
Biochemistry: 1998, 37(5);1365-75
[PubMed:9477965]
[WorldCat.org]
[DOI]
(P p)
L Wang, R Grau, M Perego, J A Hoch
A novel histidine kinase inhibitor regulating development in Bacillus subtilis.
Genes Dev: 1997, 11(19);2569-79
[PubMed:9334321]
[WorldCat.org]
[DOI]
(P p)
Y L Tzeng, J A Hoch
Molecular recognition in signal transduction: the interaction surfaces of the Spo0F response regulator with its cognate phosphorelay proteins revealed by alanine scanning mutagenesis.
J Mol Biol: 1997, 272(2);200-12
[PubMed:9299348]
[WorldCat.org]
[DOI]
(P p)
M Predich, G Nair, I Smith
Bacillus subtilis early sporulation genes kinA, spo0F, and spo0A are transcribed by the RNA polymerase containing sigma H.
J Bacteriol: 1992, 174(9);2771-8
[PubMed:1569009]
[WorldCat.org]
[DOI]
(P p)
D Burbulys, K A Trach, J A Hoch
Initiation of sporulation in B. subtilis is controlled by a multicomponent phosphorelay.
Cell: 1991, 64(3);545-52
[PubMed:1846779]
[WorldCat.org]
[DOI]
(P p)
M Perego, S P Cole, D Burbulys, K Trach, J A Hoch
Characterization of the gene for a protein kinase which phosphorylates the sporulation-regulatory proteins Spo0A and Spo0F of Bacillus subtilis.
J Bacteriol: 1989, 171(11);6187-96
[PubMed:2509430]
[WorldCat.org]
[DOI]
(P p)