Crh
Revision as of 09:15, 10 November 2011 by Czschiedrich (talk | contribs)
- Description: "Catabolite repression HPr-like protein", control of flux through the harmful methylglyoxal pathway, minor cofactor of the CcpA transcription factor
| Gene name | crh |
| Synonyms | yvcM |
| Essential | no |
| Product | catabolite repression HPr-like protein |
| Function | control of carbon flux |
| Interactions involving this protein in SubtInteract: Crh | |
| Metabolic function and regulation of this protein in SubtiPathways: Central C-metabolism | |
| MW, pI | 9,2 kDa, 4.70 |
| Gene length, protein length | 255 bp, 85 amino acids |
| Immediate neighbours | yvcN, yvcL |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 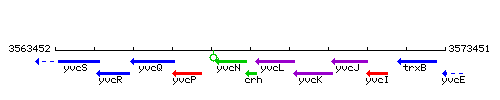
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
transcription factors and their control, phosphoproteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU34740
Phenotypes of a mutant
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: HPr family
- Paralogous protein(s): HPr
Extended information on the protein
- Kinetic information:
- Domains: HPr domain (1–85)
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure:
- UniProt: O06976
- KEGG entry: [2]
Additional information
Crh does not possess the phosphorylation site used for PTS phosphotransfer (His-15 in PtsH), it can only be phosphorylated on Ser-46
Expression and regulation
- Regulation: very weak stimuation of expression by citrate and succinate PubMed
- Regulatory mechanism:
- Additional information: Crh is weakly expressed. This results in part from a poorly conserved ribosomal binding site of the mRNA. PubMed
Biological materials
- Mutant: GP860 (aphA3), QB7097 (spc), available in Stülke lab
- Expression vector:
- lacZ fusion: see yvcI
- GFP fusion:
- two-hybrid system: crh, crh(Ser46Asp), crh(Ser46Ala) B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Görke lab
- FLAG-tag construct:
- GP69 (spc, based on pGP1331), available in the Boris Görke and Stülke labs
- pGP1200 crh(Ser46Ala) (spc, based on pGP1331), available in the Stülke lab
Labs working on this gene/protein
Boris Görke, University of Göttingen, Germany Homepage
Anne Galinier, University of Marseille, France
Wolfgang Hillen, Erlangen University, Germany Homepage
Richard Brennan, Houston, Texas, USA Homepage
Your additional remarks
References