Difference between revisions of "ClpP"
| Line 119: | Line 119: | ||
=References= | =References= | ||
| − | # Petersohn et al. (2001) Global Analysis of the General Stress Response of ''Bacillus subtilis''. | + | # Petersohn et al. (2001) Global Analysis of the General Stress Response of ''Bacillus subtilis''. ''J Bacteriol.'' '''183:''' 5617-5631 [http://www.ncbi.nlm.nih.gov/pubmed/11544224 PubMed] |
# Kock H, Gerth U, Hecker M. (2004) MurAA, catalysing the first committed step in peptidoglycan biosynthesis, is a target of Clp-dependent proteolysis in Bacillus subtilis. Mol Microbiol, 51:1087-1102. [http://www.ncbi.nlm.nih.gov/sites/entrez/14763982 PubMed] | # Kock H, Gerth U, Hecker M. (2004) MurAA, catalysing the first committed step in peptidoglycan biosynthesis, is a target of Clp-dependent proteolysis in Bacillus subtilis. Mol Microbiol, 51:1087-1102. [http://www.ncbi.nlm.nih.gov/sites/entrez/14763982 PubMed] | ||
# Gerth, U., Krüger, E., Derré, I., Msadek, T. and Hecker, M. 1998. Stress induction of the [[Bacillus subtilis clpP]] gene encoding a homologue of the proteolytic component of the ClpP protease and the involvement of ClpP and ClpX in stress tolerance. Mol. Microbiol. 28: 787-802. [http://www.ncbi.nlm.nih.gov/sites/entrez/9643546 PubMed] | # Gerth, U., Krüger, E., Derré, I., Msadek, T. and Hecker, M. 1998. Stress induction of the [[Bacillus subtilis clpP]] gene encoding a homologue of the proteolytic component of the ClpP protease and the involvement of ClpP and ClpX in stress tolerance. Mol. Microbiol. 28: 787-802. [http://www.ncbi.nlm.nih.gov/sites/entrez/9643546 PubMed] | ||
# Author1, Author2 & Author3 (year) Title ''Journal'' '''volume:''' page-page. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] | # Author1, Author2 & Author3 (year) Title ''Journal'' '''volume:''' page-page. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] | ||
Revision as of 17:18, 30 March 2009
- Description: ATP-dependent Clp protease proteolytic subunit (class III heat-shock protein)
| Gene name | clpP |
| Synonyms | yvdN |
| Essential | no |
| Product | ATP-dependent Clp protease proteolytic subunit |
| Function | protein degradation |
| MW, pI | 21 kDa, 5.008 |
| Gene length, protein length | 591 bp, 197 aa |
| Immediate neighbours | trnQ-Arg, pgcM |
| Gene sequence (+200bp) | Protein sequence |
Genetic context 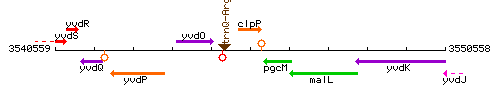
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Coordinates:
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization:
Database entries
- Structure:
- Swiss prot entry:
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
- Petersohn et al. (2001) Global Analysis of the General Stress Response of Bacillus subtilis. J Bacteriol. 183: 5617-5631 PubMed
- Kock H, Gerth U, Hecker M. (2004) MurAA, catalysing the first committed step in peptidoglycan biosynthesis, is a target of Clp-dependent proteolysis in Bacillus subtilis. Mol Microbiol, 51:1087-1102. PubMed
- Gerth, U., Krüger, E., Derré, I., Msadek, T. and Hecker, M. 1998. Stress induction of the Bacillus subtilis clpP gene encoding a homologue of the proteolytic component of the ClpP protease and the involvement of ClpP and ClpX in stress tolerance. Mol. Microbiol. 28: 787-802. PubMed
- Author1, Author2 & Author3 (year) Title Journal volume: page-page. PubMed