Difference between revisions of "AraR"
| Line 1: | Line 1: | ||
| − | * '''Description:''' repressor of the arabinose utilization genes <br/><br/> | + | * '''Description:''' transcriptional repressor ([[GntR family]], [[LacI family]]) of the arabinose utilization genes <br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
| Line 65: | Line 65: | ||
=== Additional information=== | === Additional information=== | ||
| − | |||
| − | |||
| − | |||
=The protein= | =The protein= | ||
Revision as of 17:51, 11 June 2014
- Description: transcriptional repressor (GntR family, LacI family) of the arabinose utilization genes
| Gene name | araR |
| Synonyms | araC, yvbS |
| Essential | no |
| Product | transcriptional repressor (GntR family, LacI family) |
| Function | regulation of arabinose utilization
(araABDLMNPQ-abfA) and of the araE/araR genes |
| Gene expression levels in SubtiExpress: araR
(araABDLMNPQ-abfA) and of the araE/araR genes | |
| Metabolic function and regulation of this protein in SubtiPathways: AraR | |
| MW, pI | 43 kDa, 6.341 |
| Gene length, protein length | 1152 bp, 384 aa |
| Immediate neighbours | araE, yvbT |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 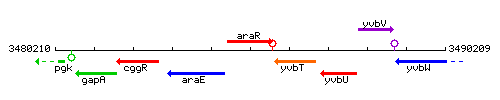
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed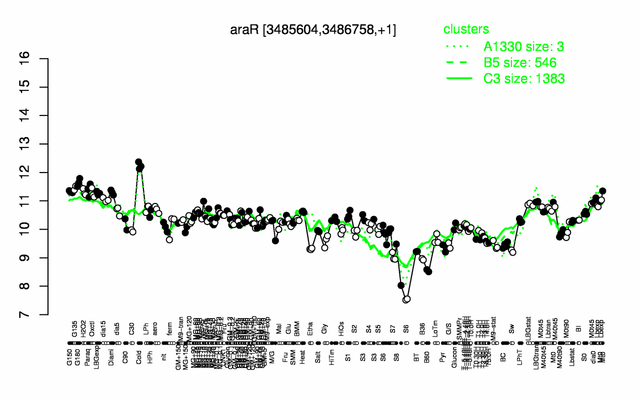
| |
Contents
Categories containing this gene/protein
utilization of specific carbon sources, transcription factors and their control
This gene is a member of the following regulons
The AraR regulon
The gene
Basic information
- Locus tag: BSU33970
Phenotypes of a mutant
Database entries
- BsubCyc: BSU33970
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: GntR family: N-terminal DNA-binding domain, LacI family: C-terminal effector-binding domain
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- N-terminal DNA-binding domain (GntR family)
- C-terminal effector-binding domain (LacI family)
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cytoplasm (according to Swiss-Prot)
Database entries
- BsubCyc: BSU33970
- UniProt: P96711
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Additional publications: PubMed