Difference between revisions of "Ggt"
(→References) |
|||
| Line 138: | Line 138: | ||
=References= | =References= | ||
| − | <pubmed>15583164,12892879,18957862,20088880 22383849 23335395,23033921,21513304,21987357 24279353 24531494 </pubmed> | + | <pubmed>15583164,12892879,18957862,20088880 22383849 23335395,23033921,21513304,21987357 24279353 24531494 24719567 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 12:06, 11 April 2014
- Description: gamma-glutamyltransferase
| Gene name | ggt |
| Synonyms | pac |
| Essential | no |
| Product | gamma-glutamyltransferase |
| Function | degradation of poly-glutamate capsules |
| Gene expression levels in SubtiExpress: ggt | |
| MW, pI | 64 kDa, 5.453 |
| Gene length, protein length | 1761 bp, 587 aa |
| Immediate neighbours | yoeD, yofA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 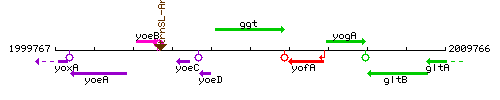
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed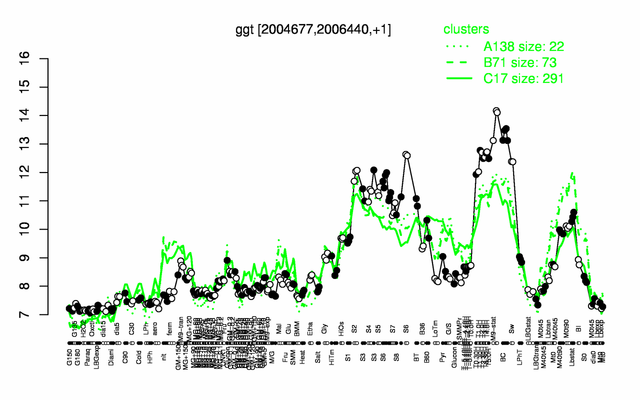
| |
Contents
Categories containing this gene/protein
capsule biosynthesis and degradation
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU18410
Phenotypes of a mutant
Database entries
- BsubCyc: BSU18410
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: (5-L-glutamyl)-peptide + an amino acid = peptide + 5-L-glutamyl amino acid (according to Swiss-Prot)
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- extracellular (signal peptide) PubMed
Database entries
- BsubCyc: BSU18410
- Structure: 2V36
- UniProt: P54422
- KEGG entry: [2]
- E.C. number: 2.3.2.2
Additional information
Expression and regulation
- Regulation:
- strongly induced in response to glucose starvation in M9 medium PubMed
- Regulatory mechanism:
- Additional information:
- translation is likely to require Efp due to the presence of several consecutive proline residues PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References