Difference between revisions of "IlvB"
(→References) |
|||
| Line 146: | Line 146: | ||
=References= | =References= | ||
| − | |||
<pubmed> 21303765 15060025,19258532,8289305,17488331,18083814, 18641142,15547269,12618455,24163341, 15547269, 12618455,12107147 1577690 12193635 15916605 15916606 15547269, 20935095 22517742 22900538 </pubmed> | <pubmed> 21303765 15060025,19258532,8289305,17488331,18083814, 18641142,15547269,12618455,24163341, 15547269, 12618455,12107147 1577690 12193635 15916605 15916606 15547269, 20935095 22517742 22900538 </pubmed> | ||
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 12:20, 22 December 2013
- Description: acetolactate synthase (large subunit)
| Gene name | ilvB |
| Synonyms | |
| Essential | no |
| Product | acetolactate synthase (large subunit) |
| Function | biosynthesis of branched-chain amino acids |
| Gene expression levels in SubtiExpress: ilvB | |
| Metabolic function and regulation of this protein in SubtiPathways: Ile, Leu, Val, Coenzyme A | |
| MW, pI | 62 kDa, 5.178 |
| Gene length, protein length | 1722 bp, 574 aa |
| Immediate neighbours | ilvH, ysnD |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 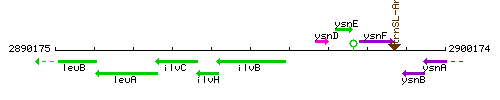
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed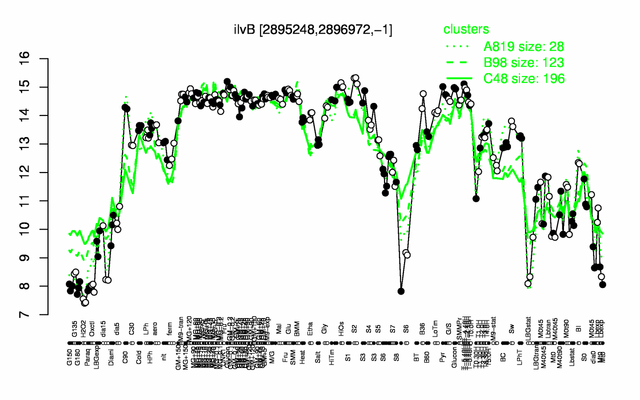
| |
Contents
The gene
Basic information
- Locus tag: BSU28310
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids, phosphoproteins
This gene is a member of the following regulons
CodY regulon, CcpA regulon, TnrA regulon, T-box
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 2 pyruvate = 2-acetolactate + CO2 (according to Swiss-Prot)
- Protein family: TPP enzyme family (according to Swiss-Prot)
Extended information on the protein
- Kinetic information:
- Modification:
- phosphorylated on Arg-297 PubMed
- Effectors of protein activity:
Database entries
- Structure: 1N53 (T box RNA)
- UniProt: P37251
- KEGG entry: [3]
- E.C. number: 2.2.1.6
Additional information
- subject to Clp-dependent proteolysis upon glucose starvation PubMed
Expression and regulation
- Regulation:
- for a complete overview on the regulation of the ilv operon, see Brinsmade et al.
- expression is stimulated in the presence of glucose (CcpA) PubMed
- repressed under nitrogen limiting conditions (glutamine or ammonium) (TnrA) PubMed
- repressed by casamino acids PubMed
- repressed during growth in the presence of branched chain amino acids (CodY) PubMed
- Regulatory mechanism:
- Additional information: subject to Clp-dependent proteolysis upon glucose starvation PubMed
Biological materials
- Mutant: GP324, ilvB under control of pXyl (cat), available in Jörg Stülke's lab
- Expression vector:
- lacZ fusion: pGP520 and pGP524 (both in pAC5), pGP523 (in pAC6), all are available in Jörg Stülke's lab
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References