Difference between revisions of "FloT"
| Line 32: | Line 32: | ||
__TOC__ | __TOC__ | ||
| − | |||
| − | |||
<br/><br/><br/><br/> | <br/><br/><br/><br/> | ||
<br/><br/><br/><br/> | <br/><br/><br/><br/> | ||
| Line 59: | Line 57: | ||
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
** delayed onset of sporulation, reduced sporulation frequency | ** delayed onset of sporulation, reduced sporulation frequency | ||
| − | ** a ''[[floT]] [[ | + | ** defect in motility {{PubMed|22753055}} |
| + | ** a ''[[floT]] [[floA]]'' double mutant does not induce [[KinC]]-dependent biofilm formation upon addition of surfactin {{PubMed|20713508}} | ||
| + | ** a ''[[floT]] [[floA]]'' double mutant has a strong synthetic defect in motility, cell morphology, and transformation efficiency {{PubMed|22753055}} | ||
=== Database entries === | === Database entries === | ||
| Line 74: | Line 74: | ||
* '''Catalyzed reaction/ biological activity:''' | * '''Catalyzed reaction/ biological activity:''' | ||
** [[SigW]]-dependent expression of ''[[fabF]]'' and the ''[[yuaF]]-[[floT]]-[[yuaI]]'' operon result in reduced membrane fluidity {{PubMed|21542858,22178969}} | ** [[SigW]]-dependent expression of ''[[fabF]]'' and the ''[[yuaF]]-[[floT]]-[[yuaI]]'' operon result in reduced membrane fluidity {{PubMed|21542858,22178969}} | ||
| + | ** recruits [[YuaF]] to focal assemblies {{PubMed|22753055}} | ||
* '''Protein family:''' | * '''Protein family:''' | ||
| Line 92: | Line 93: | ||
* '''[[SubtInteract|Interactions]]:''' | * '''[[SubtInteract|Interactions]]:''' | ||
| − | ** [[YuaF]]-[[FloT]] | + | ** [[YuaF]]-[[FloT]] {{PubMed|22753055}} |
* '''[[Localization]]:''' | * '''[[Localization]]:''' | ||
** membrane associated [http://www.ncbi.nlm.nih.gov/pubmed/18763711 PubMed] | ** membrane associated [http://www.ncbi.nlm.nih.gov/pubmed/18763711 PubMed] | ||
| − | ** co-localizes with [[ | + | ** co-localizes with [[FloA]] and [[KinC]] in discrete foci membrane {{PubMed|20713508}} |
| + | ** forms focal structure in the cytoplasma membrane {{PubMed|22753055}} | ||
=== Database entries === | === Database entries === | ||
| Line 145: | Line 147: | ||
=References= | =References= | ||
'''Additional publications:''' {{PubMed|22178969}} | '''Additional publications:''' {{PubMed|22178969}} | ||
| − | <pubmed>9987136,, 12107147, 18763711, 19383680 20713508 </pubmed> | + | <pubmed>9987136,22753055, 12107147, 18763711, 19383680 20713508 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 17:25, 6 July 2012
- Description: similar to flotillin 1, orchestration of physiological processes in lipid microdomains, involved in the control of membrane fluidity, confers (together with YuaF) resistance to cefuroxime
| Gene name | floT |
| Synonyms | yuaG, yuaH |
| Essential | no |
| Product | bacterial flotillin-like protein |
| Function | involved in the control of membrane fluidity |
| Interactions involving this protein in SubtInteract: FloT | |
| MW, pI | 55 kDa, 5.135 |
| Gene length, protein length | 1527 bp, 509 aa |
| Immediate neighbours | yuaI, yuaF |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 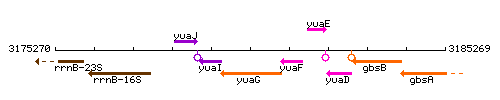
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
biofilm formation, membrane dynamics, membrane proteins sporulation/ other, cell envelope stress proteins (controlled by SigM, V, W, X, Y)
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU31010
Phenotypes of a mutant
- delayed onset of sporulation, reduced sporulation frequency
- defect in motility PubMed
- a floT floA double mutant does not induce KinC-dependent biofilm formation upon addition of surfactin PubMed
- a floT floA double mutant has a strong synthetic defect in motility, cell morphology, and transformation efficiency PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure:
- UniProt: O32076
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Additional publications: PubMed
Felix Dempwolff, Heiko M Möller, Peter L Graumann
Synthetic motility and cell shape defects associated with deletions of flotillin/reggie paralogs in Bacillus subtilis and interplay of these proteins with NfeD proteins.
J Bacteriol: 2012, 194(17);4652-61
[PubMed:22753055]
[WorldCat.org]
[DOI]
(I p)
Daniel López, Roberto Kolter
Functional microdomains in bacterial membranes.
Genes Dev: 2010, 24(17);1893-902
[PubMed:20713508]
[WorldCat.org]
[DOI]
(I p)
Catriona Donovan, Marc Bramkamp
Characterization and subcellular localization of a bacterial flotillin homologue.
Microbiology (Reading): 2009, 155(Pt 6);1786-1799
[PubMed:19383680]
[WorldCat.org]
[DOI]
(P p)
Hannes Hahne, Susanne Wolff, Michael Hecker, Dörte Becher
From complementarity to comprehensiveness--targeting the membrane proteome of growing Bacillus subtilis by divergent approaches.
Proteomics: 2008, 8(19);4123-36
[PubMed:18763711]
[WorldCat.org]
[DOI]
(I p)
Ulrike Mäder, Georg Homuth, Christian Scharf, Knut Büttner, Rüdiger Bode, Michael Hecker
Transcriptome and proteome analysis of Bacillus subtilis gene expression modulated by amino acid availability.
J Bacteriol: 2002, 184(15);4288-95
[PubMed:12107147]
[WorldCat.org]
[DOI]
(P p)
X Huang, A Gaballa, M Cao, J D Helmann
Identification of target promoters for the Bacillus subtilis extracytoplasmic function sigma factor, sigma W.
Mol Microbiol: 1999, 31(1);361-71
[PubMed:9987136]
[WorldCat.org]
[DOI]
(P p)