Difference between revisions of "Hfq"
(→Reviews) |
|||
| Line 84: | Line 84: | ||
=== Database entries === | === Database entries === | ||
| − | * '''Structure:''' [http://www.pdb.org/pdb/explore/explore.do?structureId=3HSB 3HSB] (complex with an RNA aptamer) | + | * '''Structure:''' [http://www.pdb.org/pdb/explore/explore.do?structureId=3HSB 3HSB] (complex with an RNA aptamer) {{PubMed|22053080}} |
* '''UniProt:''' [http://www.uniprot.org/uniprot/O31796 O31796] | * '''UniProt:''' [http://www.uniprot.org/uniprot/O31796 O31796] | ||
| Line 129: | Line 129: | ||
==Original publications== | ==Original publications== | ||
| − | <pubmed>12850135, 20445260 </pubmed> | + | <pubmed>12850135, 20445260 22053080 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 16:30, 6 November 2011
- Description: RNA chaperone
| Gene name | hfq |
| Synonyms | ymaH |
| Essential | no |
| Product | RNA chaperone |
| Function | unknown |
| MW, pI | 8 kDa, 8.698 |
| Gene length, protein length | 219 bp, 73 aa |
| Immediate neighbours | miaA, ymzC |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 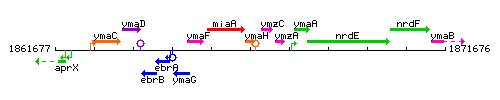
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU17340
Phenotypes of a mutant
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: hfq family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization:
Database entries
- UniProt: O31796
- KEGG entry: [2]
- E.C. number:
Additional information
Expression and regulation
- Operon:
- Regulation: repressed by glucose (7.7-fold) PubMed
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant: GP22 (cat), available in the Stülke lab
- Expression vector:
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Jörg Vogel, Ben F Luisi
Hfq and its constellation of RNA.
Nat Rev Microbiol: 2011, 9(8);578-89
[PubMed:21760622]
[WorldCat.org]
[DOI]
(I e)
Richard G Brennan, Todd M Link
Hfq structure, function and ligand binding.
Curr Opin Microbiol: 2007, 10(2);125-33
[PubMed:17395525]
[WorldCat.org]
[DOI]
(P p)
Poul Valentin-Hansen, Maiken Eriksen, Christina Udesen
The bacterial Sm-like protein Hfq: a key player in RNA transactions.
Mol Microbiol: 2004, 51(6);1525-33
[PubMed:15009882]
[WorldCat.org]
[DOI]
(P p)
Original publications