Difference between revisions of "NagZ"
(→References) |
|||
| Line 49: | Line 49: | ||
| + | |||
| + | = Categories containing this gene/protein = | ||
| + | {{SubtiWiki category|[[cell wall degradation/ turnover]]}} | ||
=The protein= | =The protein= | ||
Revision as of 18:20, 30 November 2010
- Description: N-acetylglucosaminidase
| Gene name | nagZ |
| Synonyms | yzbA, ybbD |
| Essential | no |
| Product | N-acetylglucosaminidase |
| Function | cell wall recycling |
| MW, pI | 70 kDa, 9.76 |
| Gene length, protein length | 1926 bp, 642 aa |
| Immediate neighbours | ybbC, amiE |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 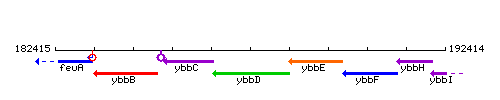
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU01660
Phenotypes of a mutant
increased autolysis PubMed
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
Categories containing this gene/protein
cell wall degradation/ turnover
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: cleaves muropeptides derived from peptidoglycan, but not peptidoglycan itself PubMed
- Protein family: glycosyl hydrolase 3 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization: secreted (with signal peptide), remains to some extent cell wall-associated PubMed
Database entries
- UniProt: P40406
- KEGG entry: [2]
- E.C. number:
Additional information
The gene is mis-annotated in KEGG as an ortholog of beta-N-acetylhexosaminidase EC 3.2.1.52. It is marked in MetaCyc as “similar to beta-hexosaminidase”. No EC annotation is available in Swiss-ProtSwiss-Prot.supporting the annotation is available. PubMed
Expression and regulation
- Operon:
- Regulation:
- expressed in late exponential and early stationary phase PubMed
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant: available in Christoph Mayer's lab PubMed
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Additional references: PubMed