Difference between revisions of "Hom"
| Line 93: | Line 93: | ||
* '''Sigma factor:''' | * '''Sigma factor:''' | ||
| − | * '''Regulation:''' | + | * '''Regulation:''' repressed by Casamino Acids [http://www.ncbi.nlm.nih.gov/pubmed/12107147 PubMed] |
* '''Regulatory mechanism:''' | * '''Regulatory mechanism:''' | ||
| Line 119: | Line 119: | ||
=References= | =References= | ||
| + | # Mäder et al. (2002) Transcriptome and Proteome Analysis of ''Bacillus subtilis'' Gene Expression Modulated by Amino Acid Availability. ''J. Bacteriol'' '''184:''' 1844288-4295 [http://www.ncbi.nlm.nih.gov/pubmed/12107147 PubMed] | ||
# Hahne et al. (2008) From complementarity to comprehensiveness - targeting the membrane proteome of growing ''Bacillus subtilis'' by divergent approaches. Proteomics '''8:''' 4123-4136 [http://www.ncbi.nlm.nih.gov/pubmed/18763711 PubMed] | # Hahne et al. (2008) From complementarity to comprehensiveness - targeting the membrane proteome of growing ''Bacillus subtilis'' by divergent approaches. Proteomics '''8:''' 4123-4136 [http://www.ncbi.nlm.nih.gov/pubmed/18763711 PubMed] | ||
# Parsot C, Cohen GN. (1988) Cloning and nucleotide sequence of the Bacillus subtilis hom gene coding for homoserine dehydrogenase. Structural and evolutionary relationships with Escherichia coli aspartokinases-homoserine dehydrogenases I and II. ''J Biol Chem.'' '''Oct 15;263(29):''' 14654-60. [http://www.ncbi.nlm.nih.gov/sites/entrez/3139660 PubMed] | # Parsot C, Cohen GN. (1988) Cloning and nucleotide sequence of the Bacillus subtilis hom gene coding for homoserine dehydrogenase. Structural and evolutionary relationships with Escherichia coli aspartokinases-homoserine dehydrogenases I and II. ''J Biol Chem.'' '''Oct 15;263(29):''' 14654-60. [http://www.ncbi.nlm.nih.gov/sites/entrez/3139660 PubMed] | ||
# Author1, Author2 & Author3 (year) Title ''Journal'' '''volume:''' page-page. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] | # Author1, Author2 & Author3 (year) Title ''Journal'' '''volume:''' page-page. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] | ||
Revision as of 11:40, 31 March 2009
- Description: homoserine dehydrogenase (NADPH)
| Gene name | hom |
| Synonyms | |
| Essential | no |
| Product | homoserine dehydrogenase (NADPH) |
| Function | biosynthesis of methionine and threonine |
| MW, pI | 47 kDa, 4.9 |
| Gene length, protein length | 1299 bp, 433 aa |
| Immediate neighbours | thrC, yutH |
| Gene sequence (+200bp) | Protein sequence |
Genetic context 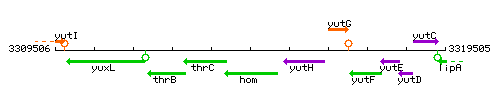
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Coordinates:
Phenotypes of a mutant
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization: membrane associated PubMed
Database entries
- Structure:
- Swiss prot entry:
- KEGG entry: [2]
- E.C. number:
Additional information
Expression and regulation
- Operon:
- Sigma factor:
- Regulation: repressed by Casamino Acids PubMed
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
- Mäder et al. (2002) Transcriptome and Proteome Analysis of Bacillus subtilis Gene Expression Modulated by Amino Acid Availability. J. Bacteriol 184: 1844288-4295 PubMed
- Hahne et al. (2008) From complementarity to comprehensiveness - targeting the membrane proteome of growing Bacillus subtilis by divergent approaches. Proteomics 8: 4123-4136 PubMed
- Parsot C, Cohen GN. (1988) Cloning and nucleotide sequence of the Bacillus subtilis hom gene coding for homoserine dehydrogenase. Structural and evolutionary relationships with Escherichia coli aspartokinases-homoserine dehydrogenases I and II. J Biol Chem. Oct 15;263(29): 14654-60. PubMed
- Author1, Author2 & Author3 (year) Title Journal volume: page-page. PubMed