Difference between revisions of "HtrB"
| Line 122: | Line 122: | ||
* '''Additional information:''' | * '''Additional information:''' | ||
** [[HtrB]] is subject to degradation by [[WprA]] and other extracellular proteases {{PubMed|24362423}} | ** [[HtrB]] is subject to degradation by [[WprA]] and other extracellular proteases {{PubMed|24362423}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium): 153 {{PubMed|24696501}} | ||
| + | ** number of protein molecules per cell (complex medium with amino acids, without glucose): 345 {{PubMed|24696501}} | ||
=Biological materials = | =Biological materials = | ||
Revision as of 09:37, 17 April 2014
- Description: membrane-anchored protein quality control protease, serine protease, response to secretion and heat stresses
| Gene name | htrB |
| Synonyms | yvtA, yvtB |
| Essential | no |
| Product | serine protease |
| Function | protein quality control |
| Gene expression levels in SubtiExpress: htrB | |
| Metabolic function and regulation of this protein in SubtiPathways: htrB | |
| MW, pI | 48 kDa, 4.632 |
| Gene length, protein length | 1374 bp, 458 aa |
| Immediate neighbours | mrgA, cssR |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 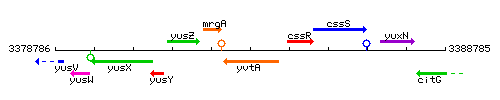
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed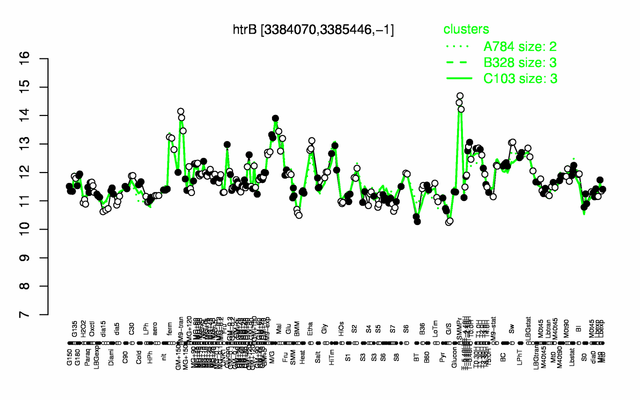
| |
Contents
Categories containing this gene/protein
proteolysis, heat shock proteins, membrane proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU33000
Phenotypes of a mutant
Database entries
- BsubCyc: BSU33000
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: peptidase S1B family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Modification:
- Effectors of protein activity:
- Localization:
- cell membrane (according to Swiss-Prot)
- randomly distributed in foci throughout the cell surface PubMed
Database entries
- BsubCyc: BSU33000
- Structure:
- UniProt: Q9R9I1
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: htrB (according to DBTBS)
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications
Laxmi Krishnappa, Carmine G Monteferrante, Jolanda Neef, Annette Dreisbach, Jan Maarten van Dijl
Degradation of extracytoplasmic catalysts for protein folding in Bacillus subtilis.
Appl Environ Microbiol: 2014, 80(4);1463-8
[PubMed:24362423]
[WorldCat.org]
[DOI]
(I p)
Susanne Pohl, Gaurav Bhavsar, Joanne Hulme, Alexandra E Bloor, Goksel Misirli, Matthew W Leckenby, David S Radford, Wendy Smith, Anil Wipat, E Diane Williamson, Colin R Harwood, Rocky M Cranenburgh
Proteomic analysis of Bacillus subtilis strains engineered for improved production of heterologous proteins.
Proteomics: 2013, 13(22);3298-308
[PubMed:24115457]
[WorldCat.org]
[DOI]
(I p)
David Noone, Eric Botella, Clodagh Butler, Annette Hansen, Inga Jende, Kevin M Devine
Signal perception by the secretion stress-responsive CssRS two-component system in Bacillus subtilis.
J Bacteriol: 2012, 194(7);1800-14
[PubMed:22307758]
[WorldCat.org]
[DOI]
(I p)
Tina Wecke, Tobias Bauer, Henning Harth, Ulrike Mäder, Thorsten Mascher
The rhamnolipid stress response of Bacillus subtilis.
FEMS Microbiol Lett: 2011, 323(2);113-23
[PubMed:22092710]
[WorldCat.org]
[DOI]
(I p)
Hanne-Leena Hyyryläinen, Milla Pietiäinen, Tuula Lundén, Anna Ekman, Marika Gardemeister, Sanna Murtomäki-Repo, Haike Antelmann, Michael Hecker, Leena Valmu, Matti Sarvas, Vesa P Kontinen
The density of negative charge in the cell wall influences two-component signal transduction in Bacillus subtilis.
Microbiology (Reading): 2007, 153(Pt 7);2126-2136
[PubMed:17600057]
[WorldCat.org]
[DOI]
(P p)
Elise Darmon, David Noone, Anne Masson, Sierd Bron, Oscar P Kuipers, Kevin M Devine, Jan Maarten van Dijl
A novel class of heat and secretion stress-responsive genes is controlled by the autoregulated CssRS two-component system of Bacillus subtilis.
J Bacteriol: 2002, 184(20);5661-71
[PubMed:12270824]
[WorldCat.org]
[DOI]
(P p)
D Noone, A Howell, R Collery, K M Devine
YkdA and YvtA, HtrA-like serine proteases in Bacillus subtilis, engage in negative autoregulation and reciprocal cross-regulation of ykdA and yvtA gene expression.
J Bacteriol: 2001, 183(2);654-63
[PubMed:11133960]
[WorldCat.org]
[DOI]
(P p)