Difference between revisions of "LexA"
(→Reviews) |
|||
| Line 60: | Line 60: | ||
=== Database entries === | === Database entries === | ||
| + | * '''BsubCyc:''' [http://bsubcyc.org/BSUB/NEW-IMAGE?type=NIL&object=BSU17850&redirect=T BSU17850] | ||
* '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/lexA.html] | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/lexA.html] | ||
| Line 94: | Line 95: | ||
=== Database entries === | === Database entries === | ||
| + | * '''BsubCyc:''' [http://bsubcyc.org/BSUB/NEW-IMAGE?type=NIL&object=BSU17850&redirect=T BSU17850] | ||
* '''Structure:''' [http://www.pdb.org/pdb/explore/explore.do?structureId=1JHH 1JHH] (from ''E. coli'', S119A mutant, 33% identity, 52% similarity) {{PubMed|11551506}} | * '''Structure:''' [http://www.pdb.org/pdb/explore/explore.do?structureId=1JHH 1JHH] (from ''E. coli'', S119A mutant, 33% identity, 52% similarity) {{PubMed|11551506}} | ||
Revision as of 13:50, 2 April 2014
- Description: transcriptional repressor of the SOS regulon
| Gene name | lexA |
| Synonyms | dinR |
| Essential | no |
| Product | transcriptional repressor of the SOS regulon |
| Function | regulation of DNA damage repair |
| Gene expression levels in SubtiExpress: lexA | |
| Function and regulation of this protein in SubtiPathways: lexA | |
| MW, pI | 22 kDa, 5.155 |
| Gene length, protein length | 615 bp, 205 aa |
| Immediate neighbours | fosB, yneA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 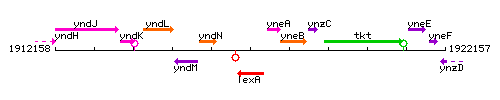
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed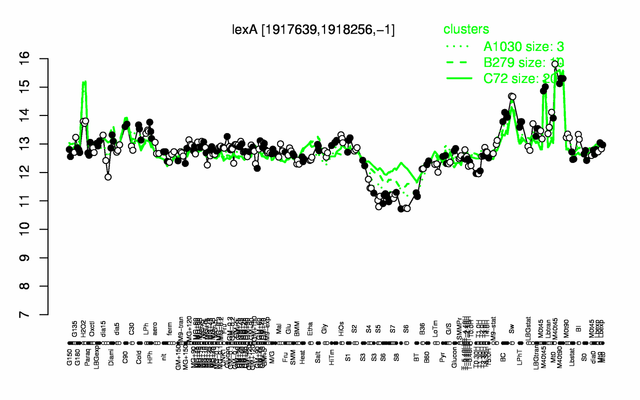
| |
Contents
Categories containing this gene/protein
DNA repair/ recombination, transcription factors and their control
This gene is a member of the following regulons
The LexA regulon
The gene
Basic information
- Locus tag: BSU17850
Phenotypes of a mutant
Database entries
- BsubCyc: BSU17850
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Represses uvrB, dinB, dinC, recA genes and itself by binding to the 14 bp palindromic sequence 5'-CGAACNNNNGTTCG-3'. In the presence of single-stranded DNA, RecA interacts with LexA causing an autocatalytic cleavage which disrupts the DNA-binding part of LexA, leading to derepression of the SOS regulon and eventually DNA repair. (according to Swiss-Prot)
- Protein family: peptidase S24 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- BsubCyc: BSU17850
- UniProt: P31080
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Sigma factor:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications