Difference between revisions of "FtsZ"
| Line 101: | Line 101: | ||
** [[FtsZ]]-[[FtsZ]] {{PubMed|17662947}} | ** [[FtsZ]]-[[FtsZ]] {{PubMed|17662947}} | ||
** [[MciZ]]-[[FtsZ]] {{PubMed|18284588}} | ** [[MciZ]]-[[FtsZ]] {{PubMed|18284588}} | ||
| − | ** [[FtsZ]] (extreme C terminus of [[FtsZ]])-[[SepF]] {{PubMed|22912848,16420366}} | + | ** [[FtsZ]] (extreme C terminus of [[FtsZ]])-[[SepF]] {{PubMed|24218584,22912848,16420366}} |
** [[RefZ]]-[[FtsZ]] {{PubMed|22730127}} | ** [[RefZ]]-[[FtsZ]] {{PubMed|22730127}} | ||
** [[FtsZ]]-[[LytE]] {{PubMed|16950129}} | ** [[FtsZ]]-[[LytE]] {{PubMed|16950129}} | ||
| Line 111: | Line 111: | ||
** septal localization partially depends on the proton motive force {{PubMed|20566861}} | ** septal localization partially depends on the proton motive force {{PubMed|20566861}} | ||
** [[Noc]] and the Min system ensure the efficient utilization of the division site at midcell in by ensuring [[FtsZ|Z ring]] placement {{PubMed|22457634}} | ** [[Noc]] and the Min system ensure the efficient utilization of the division site at midcell in by ensuring [[FtsZ|Z ring]] placement {{PubMed|22457634}} | ||
| − | ** [[FtsZ]] is anchored to the cell membrane by [[FtsA]] {{PubMed|16159787}} | + | ** [[FtsZ]] is anchored to the cell membrane by either [[FtsA]] or [[SepF]] {{PubMed|24218584,16159787}} |
=== Database entries === | === Database entries === | ||
Revision as of 17:33, 13 November 2013
- Description: cell-division initiation protein (septum formation)
| Gene name | ftsZ |
| Synonyms | ts-1 |
| Essential | yes PubMed |
| Product | cell-division initiation protein (septum formation) |
| Function | formation of Z-ring |
| Gene expression levels in SubtiExpress: ftsZ | |
| Interactions involving this protein in SubtInteract: FtsZ | |
| MW, pI | 40 kDa, 4.814 |
| Gene length, protein length | 1146 bp, 382 aa |
| Immediate neighbours | ftsA, bpr |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 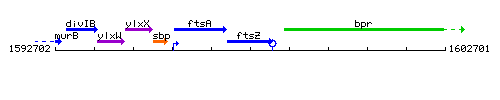
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
cell division, essential genes, membrane proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU15290
Phenotypes of a mutant
essential PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: ftsZ family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Z ring formation is inhibited upon binding of MciZ to FtsZ
- bundling of FtsZ protofilaments into strikingly long and regular tubular structures reminiscent of eukaryotic microtubules requires the prior formation of large ring polymers of SepF PubMed
- interaction with UgtP inhibits FtsZ filament formation PubMed
- FtsZ polymerization is inhibited by interaction with MinC PubMed
- Localization:
- septal at the cell membrane PubMed
- septal localization partially depends on the proton motive force PubMed
- Noc and the Min system ensure the efficient utilization of the division site at midcell in by ensuring Z ring placement PubMed
- FtsZ is anchored to the cell membrane by either FtsA or SepF PubMed
Database entries
- UniProt: P17865
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- strains:
- GP1372 (Pxyl ftsZ aphA3) disA::tet cdaS::ermC for xylose inducible expression of ftsZ, available in Jörg Stülke's lab
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody: available in the Jeff Errington lab
Labs working on this gene/protein
- Imrich Barak, Slovak Academy of Science, Bratislava, Slovakia homepage
- Leendert Hamoen, CBCB, Newcastle University, UK
Your additional remarks
References
Reviews
An-Chun Chien, Norbert S Hill, Petra Anne Levin
Cell size control in bacteria.
Curr Biol: 2012, 22(9);R340-9
[PubMed:22575476]
[WorldCat.org]
[DOI]
(I p)
Christine Kaimer, Peter L Graumann
Players between the worlds: multifunctional DNA translocases.
Curr Opin Microbiol: 2011, 14(6);719-25
[PubMed:22047950]
[WorldCat.org]
[DOI]
(I p)
Clare L Kirkpatrick, Patrick H Viollier
New(s) to the (Z-)ring.
Curr Opin Microbiol: 2011, 14(6);691-7
[PubMed:21981908]
[WorldCat.org]
[DOI]
(I p)
Harold P Erickson, David E Anderson, Masaki Osawa
FtsZ in bacterial cytokinesis: cytoskeleton and force generator all in one.
Microbiol Mol Biol Rev: 2010, 74(4);504-28
[PubMed:21119015]
[WorldCat.org]
[DOI]
(I p)
Matthew T Cabeen, Christine Jacobs-Wagner
The bacterial cytoskeleton.
Annu Rev Genet: 2010, 44;365-92
[PubMed:21047262]
[WorldCat.org]
[DOI]
(I p)
Marc Bramkamp, Suey van Baarle
Division site selection in rod-shaped bacteria.
Curr Opin Microbiol: 2009, 12(6);683-8
[PubMed:19884039]
[WorldCat.org]
[DOI]
(I p)
David W Adams, Jeff Errington
Bacterial cell division: assembly, maintenance and disassembly of the Z ring.
Nat Rev Microbiol: 2009, 7(9);642-53
[PubMed:19680248]
[WorldCat.org]
[DOI]
(I p)
Peter L Graumann
Cytoskeletal elements in bacteria.
Annu Rev Microbiol: 2007, 61;589-618
[PubMed:17506674]
[WorldCat.org]
[DOI]
(P p)
Linda A Amos, Fusinita van den Ent, Jan Löwe
Structural/functional homology between the bacterial and eukaryotic cytoskeletons.
Curr Opin Cell Biol: 2004, 16(1);24-31
[PubMed:15037301]
[WorldCat.org]
[DOI]
(P p)
FtsZ as antibacterial drug target
Filipa Marcelo, Sonia Huecas, Laura B Ruiz-Ávila, F Javier Cañada, Almudena Perona, Ana Poveda, Sonsoles Martín-Santamaría, Antonio Morreale, Jesús Jiménez-Barbero, José M Andreu
Interactions of bacterial cell division protein FtsZ with C8-substituted guanine nucleotide inhibitors. A combined NMR, biochemical and molecular modeling perspective.
J Am Chem Soc: 2013, 135(44);16418-28
[PubMed:24079270]
[WorldCat.org]
[DOI]
(I p)
Laura B Ruiz-Avila, Sonia Huecas, Marta Artola, Albert Vergoñós, Erney Ramírez-Aportela, Emilia Cercenado, Isabel Barasoain, Henar Vázquez-Villa, Mar Martín-Fontecha, Pablo Chacón, María L López-Rodríguez, José M Andreu
Synthetic inhibitors of bacterial cell division targeting the GTP-binding site of FtsZ.
ACS Chem Biol: 2013, 8(9);2072-83
[PubMed:23855511]
[WorldCat.org]
[DOI]
(I p)
Anusri Bhattacharya, Bhavya Jindal, Parminder Singh, Anindya Datta, Dulal Panda
Plumbagin inhibits cytokinesis in Bacillus subtilis by inhibiting FtsZ assembly--a mechanistic study of its antibacterial activity.
FEBS J: 2013, 280(18);4585-99
[PubMed:23841620]
[WorldCat.org]
[DOI]
(I p)
David W Adams, Ling Juan Wu, Lloyd G Czaplewski, Jeff Errington
Multiple effects of benzamide antibiotics on FtsZ function.
Mol Microbiol: 2011, 80(1);68-84
[PubMed:21276094]
[WorldCat.org]
[DOI]
(I p)
Simranjeet Kaur, Niraj H Modi, Dulal Panda, Nilanjan Roy
Probing the binding site of curcumin in Escherichia coli and Bacillus subtilis FtsZ--a structural insight to unveil antibacterial activity of curcumin.
Eur J Med Chem: 2010, 45(9);4209-14
[PubMed:20615583]
[WorldCat.org]
[DOI]
(I p)
Kumiko W Shimotohno, Fujio Kawamura, Yousuke Natori, Hideaki Nanamiya, Junji Magae, Hiromitsu Ogata, Toyoshige Endo, Takeshi Suzuki, Hiroshi Yamaki
Inhibition of septation in Bacillus subtilis by a peptide antibiotic, edeine B(1).
Biol Pharm Bull: 2010, 33(4);568-71
[PubMed:20410587]
[WorldCat.org]
[DOI]
(I p)
José M Andreu, Claudia Schaffner-Barbero, Sonia Huecas, Dulce Alonso, María L Lopez-Rodriguez, Laura B Ruiz-Avila, Rafael Núñez-Ramírez, Oscar Llorca, Antonio J Martín-Galiano
The antibacterial cell division inhibitor PC190723 is an FtsZ polymer-stabilizing agent that induces filament assembly and condensation.
J Biol Chem: 2010, 285(19);14239-46
[PubMed:20212044]
[WorldCat.org]
[DOI]
(I p)
Tushar K Beuria, Parminder Singh, Avadhesha Surolia, Dulal Panda
Promoting assembly and bundling of FtsZ as a strategy to inhibit bacterial cell division: a new approach for developing novel antibacterial drugs.
Biochem J: 2009, 423(1);61-9
[PubMed:19583568]
[WorldCat.org]
[DOI]
(I e)
Neil R Stokes, Jörg Sievers, Stephanie Barker, James M Bennett, David R Brown, Ian Collins, Veronica M Errington, David Foulger, Michelle Hall, Rowena Halsey, Hazel Johnson, Valerie Rose, Helena B Thomaides, David J Haydon, Lloyd G Czaplewski, Jeff Errington
Novel inhibitors of bacterial cytokinesis identified by a cell-based antibiotic screening assay.
J Biol Chem: 2005, 280(48);39709-15
[PubMed:16174771]
[WorldCat.org]
[DOI]
(P p)
Other original Publications
Bhavya Jindal, Dulal Panda
Understanding FtsZ assembly: cues from the behavior of its N- and C-terminal domains.
Biochemistry: 2013, 52(40);7071-81
[PubMed:24007276]
[WorldCat.org]
[DOI]
(I p)
David Ballesteros-Plaza, Isabel Holguera, Dirk-Jan Scheffers, Margarita Salas, Daniel Muñoz-Espín
Phage 29 phi protein p1 promotes replication by associating with the FtsZ ring of the divisome in Bacillus subtilis.
Proc Natl Acad Sci U S A: 2013, 110(30);12313-8
[PubMed:23836667]
[WorldCat.org]
[DOI]
(I p)
Erik Nico Trip, Jan-Willem Veening, Eric J Stewart, Jeff Errington, Dirk-Jan Scheffers
Balanced transcription of cell division genes in Bacillus subtilis as revealed by single cell analysis.
Environ Microbiol: 2013, 15(12);3196-209
[PubMed:23701187]
[WorldCat.org]
[DOI]
(I p)
P J Buske, Petra Anne Levin
A flexible C-terminal linker is required for proper FtsZ assembly in vitro and cytokinetic ring formation in vivo.
Mol Microbiol: 2013, 89(2);249-63
[PubMed:23692518]
[WorldCat.org]
[DOI]
(I p)
Valdir Blasios, Alexandre W Bisson-Filho, Patricia Castellen, Maria Luiza C Nogueira, Jefferson Bettini, Rodrigo V Portugal, Ana Carolina M Zeri, Frederico J Gueiros-Filho
Genetic and biochemical characterization of the MinC-FtsZ interaction in Bacillus subtilis.
PLoS One: 2013, 8(4);e60690
[PubMed:23577149]
[WorldCat.org]
[DOI]
(I p)
Michael P Strauss, Andrew T F Liew, Lynne Turnbull, Cynthia B Whitchurch, Leigh G Monahan, Elizabeth J Harry
3D-SIM super resolution microscopy reveals a bead-like arrangement for FtsZ and the division machinery: implications for triggering cytokinesis.
PLoS Biol: 2012, 10(9);e1001389
[PubMed:22984350]
[WorldCat.org]
[DOI]
(I p)
An-Chun Chien, Shannon Kian Gharabiklou Zareh, Yan Mei Wang, Petra Anne Levin
Changes in the oligomerization potential of the division inhibitor UgtP co-ordinate Bacillus subtilis cell size with nutrient availability.
Mol Microbiol: 2012, 86(3);594-610
[PubMed:22931116]
[WorldCat.org]
[DOI]
(I p)
Ewa Cendrowicz, Sebastiaan P van Kessel, Laura S van Bezouwen, Neeraj Kumar, Egbert J Boekema, Dirk-Jan Scheffers
Bacillus subtilis SepF binds to the C-terminus of FtsZ.
PLoS One: 2012, 7(8);e43293
[PubMed:22912848]
[WorldCat.org]
[DOI]
(I p)
Jennifer K Wagner-Herman, Remi Bernard, Roisin Dunne, Alexandre W Bisson-Filho, Krithika Kumar, Trang Nguyen, Lawrence Mulcahy, John Koullias, Frederico J Gueiros-Filho, David Z Rudner
RefZ facilitates the switch from medial to polar division during spore formation in Bacillus subtilis.
J Bacteriol: 2012, 194(17);4608-18
[PubMed:22730127]
[WorldCat.org]
[DOI]
(I p)
Christopher D A Rodrigues, Elizabeth J Harry
The Min system and nucleoid occlusion are not required for identifying the division site in Bacillus subtilis but ensure its efficient utilization.
PLoS Genet: 2012, 8(3);e1002561
[PubMed:22457634]
[WorldCat.org]
[DOI]
(I p)
Paul J Buske, Petra Anne Levin
Extreme C terminus of bacterial cytoskeletal protein FtsZ plays fundamental role in assembly independent of modulatory proteins.
J Biol Chem: 2012, 287(14);10945-57
[PubMed:22298780]
[WorldCat.org]
[DOI]
(I p)
Muhammet E Gündoğdu, Yoshikazu Kawai, Nada Pavlendova, Naotake Ogasawara, Jeff Errington, Dirk-Jan Scheffers, Leendert W Hamoen
Large ring polymers align FtsZ polymers for normal septum formation.
EMBO J: 2011, 30(3);617-26
[PubMed:21224850]
[WorldCat.org]
[DOI]
(I p)
Phoebe C Jennings, Guy C Cox, Leigh G Monahan, Elizabeth J Harry
Super-resolution imaging of the bacterial cytokinetic protein FtsZ.
Micron: 2011, 42(4);336-41
[PubMed:20933427]
[WorldCat.org]
[DOI]
(I p)
Remi Bernard, Kathleen A Marquis, David Z Rudner
Nucleoid occlusion prevents cell division during replication fork arrest in Bacillus subtilis.
Mol Microbiol: 2010, 78(4);866-82
[PubMed:20807205]
[WorldCat.org]
[DOI]
(I p)
Inês Filipa Fernandes de Oliveira, Anabela de Sousa Borges, Viola Kooij, Jeremy Bartosiak-Jentys, Joen Luirink, Dirk-Jan Scheffers
Characterization of ftsZ mutations that render Bacillus subtilis resistant to MinC.
PLoS One: 2010, 5(8);e12048
[PubMed:20711458]
[WorldCat.org]
[DOI]
(I e)
Henrik Strahl, Leendert W Hamoen
Membrane potential is important for bacterial cell division.
Proc Natl Acad Sci U S A: 2010, 107(27);12281-6
[PubMed:20566861]
[WorldCat.org]
[DOI]
(I p)
S Moriya, R A Rashid, C D Andrade Rodrigues, E J Harry
Influence of the nucleoid and the early stages of DNA replication on positioning the division site in Bacillus subtilis.
Mol Microbiol: 2010, 76(3);634-47
[PubMed:20199598]
[WorldCat.org]
[DOI]
(I p)
Leigh G Monahan, Andrew Robinson, Elizabeth J Harry
Lateral FtsZ association and the assembly of the cytokinetic Z ring in bacteria.
Mol Microbiol: 2009, 74(4);1004-17
[PubMed:19843223]
[WorldCat.org]
[DOI]
(I p)
Pamela Gamba, Jan-Willem Veening, Nigel J Saunders, Leendert W Hamoen, Richard A Daniel
Two-step assembly dynamics of the Bacillus subtilis divisome.
J Bacteriol: 2009, 191(13);4186-94
[PubMed:19429628]
[WorldCat.org]
[DOI]
(I p)
M Leaver, P Domínguez-Cuevas, J M Coxhead, R A Daniel, J Errington
Life without a wall or division machine in Bacillus subtilis.
Nature: 2009, 457(7231);849-53
[PubMed:19212404]
[WorldCat.org]
[DOI]
(I p)
James A Gregory, Eric C Becker, Kit Pogliano
Bacillus subtilis MinC destabilizes FtsZ-rings at new cell poles and contributes to the timing of cell division.
Genes Dev: 2008, 22(24);3475-88
[PubMed:19141479]
[WorldCat.org]
[DOI]
(P p)
Daniel P Haeusser, Amy H Lee, Richard B Weart, Petra Anne Levin
ClpX inhibits FtsZ assembly in a manner that does not require its ATP hydrolysis-dependent chaperone activity.
J Bacteriol: 2009, 191(6);1986-91
[PubMed:19136590]
[WorldCat.org]
[DOI]
(I p)
Dirk-Jan Scheffers
The effect of MinC on FtsZ polymerization is pH dependent and can be counteracted by ZapA.
FEBS Lett: 2008, 582(17);2601-8
[PubMed:18588879]
[WorldCat.org]
[DOI]
(P p)
Aaron A Handler, Joo Eun Lim, Richard Losick
Peptide inhibitor of cytokinesis during sporulation in Bacillus subtilis.
Mol Microbiol: 2008, 68(3);588-99
[PubMed:18284588]
[WorldCat.org]
[DOI]
(I p)
Richard B Weart, Amy H Lee, An-Chun Chien, Daniel P Haeusser, Norbert S Hill, Petra Anne Levin
A metabolic sensor governing cell size in bacteria.
Cell: 2007, 130(2);335-47
[PubMed:17662947]
[WorldCat.org]
[DOI]
(P p)
Rut Carballido-López, Alex Formstone, Ying Li, S Dusko Ehrlich, Philippe Noirot, Jeff Errington
Actin homolog MreBH governs cell morphogenesis by localization of the cell wall hydrolase LytE.
Dev Cell: 2006, 11(3);399-409
[PubMed:16950129]
[WorldCat.org]
[DOI]
(P p)
Shu Ishikawa, Yoshikazu Kawai, Konosuke Hiramatsu, Masayoshi Kuwano, Naotake Ogasawara
A new FtsZ-interacting protein, YlmF, complements the activity of FtsA during progression of cell division in Bacillus subtilis.
Mol Microbiol: 2006, 60(6);1364-80
[PubMed:16796675]
[WorldCat.org]
[DOI]
(P p)
Katherine A Michie, Leigh G Monahan, Peter L Beech, Elizabeth J Harry
Trapping of a spiral-like intermediate of the bacterial cytokinetic protein FtsZ.
J Bacteriol: 2006, 188(5);1680-90
[PubMed:16484179]
[WorldCat.org]
[DOI]
(P p)
Leendert W Hamoen, Jean-Christophe Meile, Wouter de Jong, Philippe Noirot, Jeff Errington
SepF, a novel FtsZ-interacting protein required for a late step in cell division.
Mol Microbiol: 2006, 59(3);989-99
[PubMed:16420366]
[WorldCat.org]
[DOI]
(P p)
S O Jensen, L S Thompson, E J Harry
Cell division in Bacillus subtilis: FtsZ and FtsA association is Z-ring independent, and FtsA is required for efficient midcell Z-Ring assembly.
J Bacteriol: 2005, 187(18);6536-44
[PubMed:16159787]
[WorldCat.org]
[DOI]
(P p)
Richard B Weart, Shunji Nakano, Brooke E Lane, Peter Zuber, Petra Anne Levin
The ClpX chaperone modulates assembly of the tubulin-like protein FtsZ.
Mol Microbiol: 2005, 57(1);238-49
[PubMed:15948963]
[WorldCat.org]
[DOI]
(P p)
Andrea Feucht, Jeffery Errington
ftsZ mutations affecting cell division frequency, placement and morphology in Bacillus subtilis.
Microbiology (Reading): 2005, 151(Pt 6);2053-2064
[PubMed:15942012]
[WorldCat.org]
[DOI]
(P p)
David E Anderson, Frederico J Gueiros-Filho, Harold P Erickson
Assembly dynamics of FtsZ rings in Bacillus subtilis and Escherichia coli and effects of FtsZ-regulating proteins.
J Bacteriol: 2004, 186(17);5775-81
[PubMed:15317782]
[WorldCat.org]
[DOI]
(P p)
Harry H Low, Martin C Moncrieffe, Jan Löwe
The crystal structure of ZapA and its modulation of FtsZ polymerisation.
J Mol Biol: 2004, 341(3);839-52
[PubMed:15288790]
[WorldCat.org]
[DOI]
(P p)
Richard B Weart, Petra Anne Levin
Growth rate-dependent regulation of medial FtsZ ring formation.
J Bacteriol: 2003, 185(9);2826-34
[PubMed:12700262]
[WorldCat.org]
[DOI]
(P p)
Frederico J Gueiros-Filho, Richard Losick
A widely conserved bacterial cell division protein that promotes assembly of the tubulin-like protein FtsZ.
Genes Dev: 2002, 16(19);2544-56
[PubMed:12368265]
[WorldCat.org]
[DOI]
(P p)
Andrea Feucht, Laura Abbotts, Jeffery Errington
The cell differentiation protein SpoIIE contains a regulatory site that controls its phosphatase activity in response to asymmetric septation.
Mol Microbiol: 2002, 45(4);1119-30
[PubMed:12180929]
[WorldCat.org]
[DOI]
(P p)
Sigal Ben-Yehuda, Richard Losick
Asymmetric cell division in B. subtilis involves a spiral-like intermediate of the cytokinetic protein FtsZ.
Cell: 2002, 109(2);257-66
[PubMed:12007411]
[WorldCat.org]
[DOI]
(P p)
P L Graumann, R Losick
Coupling of asymmetric division to polar placement of replication origin regions in Bacillus subtilis.
J Bacteriol: 2001, 183(13);4052-60
[PubMed:11395470]
[WorldCat.org]
[DOI]
(P p)
Keisuke Fukuchi, Yasuhiro Kasahara, Kei Asai, Kazuo Kobayashi, Shigeki Moriya, Naotake Ogasawara
The essential two-component regulatory system encoded by yycF and yycG modulates expression of the ftsAZ operon in Bacillus subtilis.
Microbiology (Reading): 2000, 146 ( Pt 7);1573-1583
[PubMed:10878122]
[WorldCat.org]
[DOI]
(P p)
I Lucet, A Feucht, M D Yudkin, J Errington
Direct interaction between the cell division protein FtsZ and the cell differentiation protein SpoIIE.
EMBO J: 2000, 19(7);1467-75
[PubMed:10747015]
[WorldCat.org]
[DOI]
(P p)
P A Levin, I G Kurtser, A D Grossman
Identification and characterization of a negative regulator of FtsZ ring formation in Bacillus subtilis.
Proc Natl Acad Sci U S A: 1999, 96(17);9642-7
[PubMed:10449747]
[WorldCat.org]
[DOI]
(P p)
N King, O Dreesen, P Stragier, K Pogliano, R Losick
Septation, dephosphorylation, and the activation of sigmaF during sporulation in Bacillus subtilis.
Genes Dev: 1999, 13(9);1156-67
[PubMed:10323866]
[WorldCat.org]
[DOI]
(P p)
L B Pedersen, E R Angert, P Setlow
Septal localization of penicillin-binding protein 1 in Bacillus subtilis.
J Bacteriol: 1999, 181(10);3201-11
[PubMed:10322023]
[WorldCat.org]
[DOI]
(P p)
A Khvorova, L Zhang, M L Higgins, P J Piggot
The spoIIE locus is involved in the Spo0A-dependent switch in the location of FtsZ rings in Bacillus subtilis.
J Bacteriol: 1998, 180(5);1256-60
[PubMed:9495766]
[WorldCat.org]
[DOI]
(P p)
P A Levin, R Losick, P Stragier, F Arigoni
Localization of the sporulation protein SpoIIE in Bacillus subtilis is dependent upon the cell division protein FtsZ.
Mol Microbiol: 1997, 25(5);839-46
[PubMed:9364910]
[WorldCat.org]
[DOI]
(P p)
X Wang, J Huang, A Mukherjee, C Cao, J Lutkenhaus
Analysis of the interaction of FtsZ with itself, GTP, and FtsA.
J Bacteriol: 1997, 179(17);5551-9
[PubMed:9287012]
[WorldCat.org]
[DOI]
(P p)
P A Levin, R Losick
Transcription factor Spo0A switches the localization of the cell division protein FtsZ from a medial to a bipolar pattern in Bacillus subtilis.
Genes Dev: 1996, 10(4);478-88
[PubMed:8600030]
[WorldCat.org]
[DOI]
(P p)
M A Strauch
Delineation of AbrB-binding sites on the Bacillus subtilis spo0H, kinB, ftsAZ, and pbpE promoters and use of a derived homology to identify a previously unsuspected binding site in the bsuB1 methylase promote.
J Bacteriol: 1995, 177(23);6999-7002
[PubMed:7592498]
[WorldCat.org]
[DOI]
(P p)
G Gonzy-Tréboul, C Karmazyn-Campelli, P Stragier
Developmental regulation of transcription of the Bacillus subtilis ftsAZ operon.
J Mol Biol: 1992, 224(4);967-79
[PubMed:1569582]
[WorldCat.org]
[DOI]
(P p)