Difference between revisions of "FeuA"
(→Database entries) |
|||
| Line 78: | Line 78: | ||
=== Database entries === | === Database entries === | ||
| − | * '''Structure:''' [http://www.rcsb.org/pdb/explore.do?structureId=2PHZ 2PHZ], [http://www.rcsb.org/pdb/explore.do?structureId=2WHY 2WHY] (complex with ferri-bacillibactin) | + | * '''Structure:''' [http://www.rcsb.org/pdb/explore.do?structureId=2PHZ 2PHZ], [http://www.rcsb.org/pdb/explore.do?structureId=2WI8 2WI8], [http://www.rcsb.org/pdb/explore.do?structureId=2WHY 2WHY] (complex with ferri-bacillibactin) |
* '''UniProt:''' [http://www.uniprot.org/uniprot/P40409 P40409] | * '''UniProt:''' [http://www.uniprot.org/uniprot/P40409 P40409] | ||
| Line 125: | Line 125: | ||
=References= | =References= | ||
| − | <pubmed>14563870,16672620,10092453,16889643,17725565,12354229,18957862 18763711, </pubmed> | + | <pubmed>19746494,14563870,16672620,10092453,16889643,17725565,12354229,18957862 18763711, </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 09:54, 14 October 2009
- Description: ABC-transporter for the siderophores enterobactin and bacillibactin (binding protein), with YusV as ATPase
| Gene name | feuA |
| Synonyms | |
| Essential | no |
| Product | ABC-transporter for the siderophores enterobactin and bacillibactin (binding protein) |
| Function | iron acquisition |
| Metabolic function and regulation of this protein in SubtiPathways: Stress | |
| MW, pI | 34 kDa, 8.018 |
| Gene length, protein length | 951 bp, 317 aa |
| Immediate neighbours | feuB, btr |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 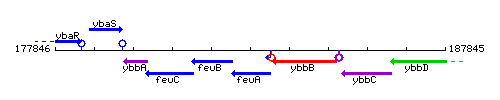
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU01630
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: Fe/B12 periplasmic-binding domain (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization: cell membrane (according to Swiss-Prot), extracellular (signal peptide) PubMed, membrane PubMed
Database entries
- UniProt: P40409
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References