Difference between revisions of "AcoC"
| Line 82: | Line 82: | ||
* '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/O31550 O31550] | * '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/O31550 O31550] | ||
| − | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu | + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu:BSU08080] |
* '''E.C. number:''' [http://www.expasy.org/enzyme/2.3.1.12 2.3.1.12] | * '''E.C. number:''' [http://www.expasy.org/enzyme/2.3.1.12 2.3.1.12] | ||
Revision as of 00:59, 25 June 2009
- Description: acetoin dehydrogenase E2 component (dihydrolipoamide acetyltransferase)
| Gene name | acoC |
| Synonyms | yfjI |
| Essential | no |
| Product | acetoin dehydrogenase E2 component (dihydrolipoamide acetyltransferase) |
| Function | acetoin utilization |
| Metabolic function and regulation of this protein in SubtiPathways: Central C-metabolism | |
| MW, pI | 42 kDa, 6.524 |
| Gene length, protein length | 1194 bp, 398 aa |
| Immediate neighbours | acoB, acoL |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 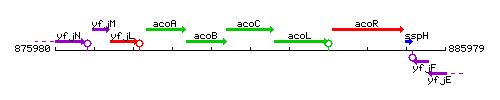
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU08080
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Acetyl-CoA + enzyme N(6)-(dihydrolipoyl)lysine = CoA + enzyme N(6)-(S-acetyldihydrolipoyl)lysine (according to Swiss-Prot)
- Protein family: lipoyl-binding domain (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization: Membrane-proximal (Spotty) PubMed
Database entries
- Structure:
- Swiss prot entry: O31550
- KEGG entry: [3]
- E.C. number: 2.3.1.12
Additional information
Expression and regulation
- Regulatory mechanism: CcpA: transcription repression, AcoR: transcription activation (interaction with SigL-containing RNA polymerase) PubMed
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Michel Debarbouille, Pasteur Institute, Paris, France Homepage
Your additional remarks
References