Difference between revisions of "GltB"
(→Biological materials) |
|||
| Line 130: | Line 130: | ||
=Biological materials = | =Biological materials = | ||
| − | * '''Mutant:''' GP807 (del '' | + | * '''Mutant:''' |
| + | ** GP807 (del ''[[gltA]]-[[gltB]]''::''tet'') , available in [[Jörg Stülke]]'s lab | ||
| + | ** GP517 (ermC), available in [[Jörg Stülke]]'s lab | ||
* '''Expression vector:''' | * '''Expression vector:''' | ||
| − | ** pGP1119 (in [[pGP380]], for SPINE, expression in ''B. subtilis''), available in [[Stülke]] lab | + | ** pGP1119 (in [[pGP380]], for SPINE, expression in ''B. subtilis''), available in [[Jörg Stülke]]'s lab |
* '''lacZ fusion:''' see ''[[gltA]]'' | * '''lacZ fusion:''' see ''[[gltA]]'' | ||
| Line 139: | Line 141: | ||
* '''GFP fusion:''' | * '''GFP fusion:''' | ||
| − | * '''two-hybrid system:''' ''B. pertussis'' adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Stülke]] lab | + | * '''two-hybrid system:''' ''B. pertussis'' adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Jörg Stülke]]'s lab |
* '''Antibody:''' | * '''Antibody:''' | ||
Revision as of 08:13, 24 September 2014
- Description: small subunit of glutamate synthase
| Gene name | gltB |
| Synonyms | |
| Essential | no |
| Product | glutamate synthase (small subunit) |
| Function | glutamate biosynthesis |
| Gene expression levels in SubtiExpress: gltB | |
| Interactions involving this protein in SubtInteract: GltB | |
| Metabolic function and regulation of this protein in SubtiPathways: gltB | |
| MW, pI | 54.6 kDa, 7.69 |
| Gene length, protein length | 1479 bp, 493 amino acids |
| Immediate neighbours | yogA, gltA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 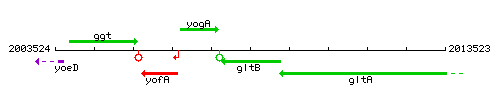
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed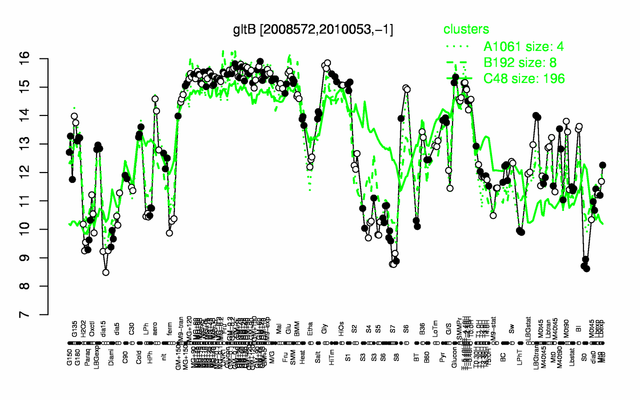
| |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids, glutamate metabolism
This gene is a member of the following regulons
GltC regulon, FsrA regulon, TnrA regulon
The gene
Basic information
- Locus tag: BSU18440
Phenotypes of a mutant
auxotrophic for glutamate
Database entries
- BsubCyc: BSU18440
- DBTBS entry: [1]
- SubtiList entry:[2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 2 L-glutamate + NADP+ = L-glutamine + 2-oxoglutarate + NADPH (according to Swiss-Prot) 2 L-glutamate + NADP(+) <=> L-glutamine + 2-oxoglutarate + NADPH
- Protein family: glutamate synthase family.
- Paralogous protein(s): none
Extended information on the protein
- Kinetic information:
- Domains:
- nucleotide binding domain (NADP) (299–313)
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cytoplasm (according to Swiss-Prot)
Database entries
- BsubCyc: BSU18440
- UniProt: O34399
- KEGG entry: [3]
- E.C. number: 1.4.1.13
Additional information
Expression and regulation
- Regulation: see gltA
- Additional information:
- number of protein molecules per cell (minimal medium with glucose and ammonium): 1128 PubMed
Biological materials
- Mutant:
- GP807 (del gltA-gltB::tet) , available in Jörg Stülke's lab
- GP517 (ermC), available in Jörg Stülke's lab
- Expression vector:
- pGP1119 (in pGP380, for SPINE, expression in B. subtilis), available in Jörg Stülke's lab
- lacZ fusion: see gltA
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Jörg Stülke's lab
- Antibody:
Labs working on this gene/protein
Linc Sonenshein, Tufts University, Boston, MA, USA Homepage
Jörg Stülke, University of Göttingen, Germany Homepage
Fabian Commichau University of Göttingen, Germany Homepage
Your additional remarks
References
Reviews
Original publications