Difference between revisions of "Zwf"
| Line 120: | Line 120: | ||
* '''Additional information:''' | * '''Additional information:''' | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium): 2681 {{PubMed|24696501}} | ||
| + | ** number of protein molecules per cell (complex medium with amino acids, without glucose): 3601 {{PubMed|24696501}} | ||
=Biological materials = | =Biological materials = | ||
Revision as of 09:29, 17 April 2014
- Description: glucose 6-phosphate dehydrogenase, pentose-phosphate pathway
| Gene name | zwf |
| Synonyms | yqjJ |
| Essential | no |
| Product | glucose 6-phosphate dehydrogenase |
| Function | initiation of the pentose phosphate pathway |
| Gene expression levels in SubtiExpress: zwf | |
| Metabolic function and regulation of this protein in SubtiPathways: zwf | |
| MW, pI | 55 kDa, 5.28 |
| Gene length, protein length | 1467 bp, 489 aa |
| Immediate neighbours | rnz, gndA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 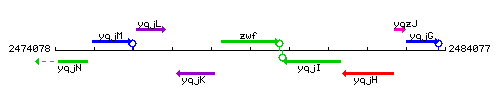
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU23850
Phenotypes of a mutant
Database entries
- BsubCyc: BSU23850
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: D-glucose 6-phosphate + NADP+ = D-glucono-1,5-lactone 6-phosphate + NADPH (according to Swiss-Prot)
- Protein family: glucose-6-phosphate dehydrogenase family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Modification:
- Cofactors: Mg2+, Mn2+, Ca2+
- Effectors of protein activity:
- NAD+, NADP+ and NADPH affect the enzyme kinetic PubMed
Database entries
- BsubCyc: BSU23850
- UniProt: P54547
- KEGG entry: [3]
- E.C. number: 1.1.1.49
Additional information
The enzyme is a dimer PubMed
Expression and regulation
- Operon: zwf PubMed
- Regulation: constitutive PubMed
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- SM-NB3 (zwf-spc), available in Anne Galinier's and Boris Görke's labs
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Michael Kohlstedt, Praveen K Sappa, Hanna Meyer, Sandra Maaß, Adrienne Zaprasis, Tamara Hoffmann, Judith Becker, Leif Steil, Michael Hecker, Jan Maarten van Dijl, Michael Lalk, Ulrike Mäder, Jörg Stülke, Erhard Bremer, Uwe Völker, Christoph Wittmann
Adaptation of Bacillus subtilis carbon core metabolism to simultaneous nutrient limitation and osmotic challenge: a multi-omics perspective.
Environ Microbiol: 2014, 16(6);1898-917
[PubMed:24571712]
[WorldCat.org]
[DOI]
(I p)
Yun Xia Duan, Tao Chen, Xun Chen, Xue Ming Zhao
Overexpression of glucose-6-phosphate dehydrogenase enhances riboflavin production in Bacillus subtilis.
Appl Microbiol Biotechnol: 2010, 85(6);1907-14
[PubMed:19779711]
[WorldCat.org]
[DOI]
(I p)
Hans-Matti Blencke, Georg Homuth, Holger Ludwig, Ulrike Mäder, Michael Hecker, Jörg Stülke
Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways.
Metab Eng: 2003, 5(2);133-49
[PubMed:12850135]
[WorldCat.org]
[DOI]
(P p)
P Rowland, A K Basak, S Gover, H R Levy, M J Adams
The three-dimensional structure of glucose 6-phosphate dehydrogenase from Leuconostoc mesenteroides refined at 2.0 A resolution.
Structure: 1994, 2(11);1073-87
[PubMed:7881907]
[WorldCat.org]
[DOI]
(P p)
S Ujita, K Kimura
Glucose-6-phosphate dehydrogenase, vegetative and spore Bacillus subtilis.
Methods Enzymol: 1982, 89 Pt D;258-61
[PubMed:6292660]
[WorldCat.org]
[DOI]
(P p)