Difference between revisions of "DltA"
| Line 1: | Line 1: | ||
| − | * '''Description:''' D-alanyl-D-alanine carrier protein ligase, alanylation of | + | * '''Description:''' D-alanyl-D-alanine carrier protein ligase, alanylation of teichoic acid provides some resistance against positively charged antimicrobial peptides <br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
Revision as of 13:54, 31 October 2012
- Description: D-alanyl-D-alanine carrier protein ligase, alanylation of teichoic acid provides some resistance against positively charged antimicrobial peptides
| Gene name | dltA |
| Synonyms | ipa-5r, dae |
| Essential | no |
| Product | D-alanyl-D-alanine carrier protein ligase |
| Function | biosynthesis of teichoic acid |
| Gene expression levels in SubtiExpress: dltA | |
| MW, pI | 55 kDa, 4.929 |
| Gene length, protein length | 1509 bp, 503 aa |
| Immediate neighbours | menA, dltB |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 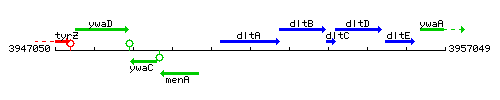
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
cell wall synthesis, biosynthesis of cell wall components, cell envelope stress proteins (controlled by SigM, V, W, X, Y)
This gene is a member of the following regulons
SigD regulon, SigM regulon, SigX regulon, Spo0A regulon, stringent response, YvrHb regulon
The gene
Basic information
- Locus tag: BSU38500
Phenotypes of a mutant
- increased sensitivity to lysozyme PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + D-alanine + poly(ribitol phosphate) = AMP + diphosphate + O-D-alanyl-poly(ribitol phosphate) (according to Swiss-Prot)
- Protein family: DltA subfamily (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- cytoplasm (according to Swiss-Prot)
Database entries
- Structure: 3E7X
- UniProt: P39581
- KEGG entry: [3]
- E.C. number: 6.1.1.13
Additional information
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Mohamed Marahiel, Marburg University, Germany homepage
Your additional remarks
References
Additional publications: PubMed