Difference between revisions of "MtnD"
(→References) |
|||
| Line 122: | Line 122: | ||
=References= | =References= | ||
| − | <pubmed>12022921,11914366,,12107147, 15102328 </pubmed> | + | <pubmed>12022921,11914366,,12107147, 15102328 12787499 18039762 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 06:33, 24 February 2010
- Description: 1,2,-dihydroxy-3-keto-5-methylthiopentene dioxygenase
| Gene name | mtnD |
| Synonyms | ykrZ |
| Essential | no |
| Product | 1,2,-dihydroxy-3-keto-5-methylthiopentene dioxygenase |
| Function | methionine salvage |
| Metabolic function and regulation of this protein in SubtiPathways: Cys, Met & Sulfate assimilation | |
| MW, pI | 20 kDa, 4.408 |
| Gene length, protein length | 534 bp, 178 aa |
| Immediate neighbours | mtnB, ykvA |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 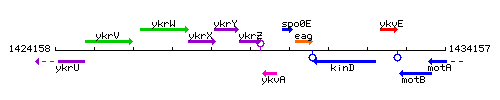
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU13620
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 1,2-dihydroxy-5-(methylthio)pent-1-en-3-one + O2 = 3-(methylthio)propanoate + formate + CO (according to Swiss-Prot)
- Protein family: acireductone dioxygenase (ARD) family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization:
Database entries
- Structure:
- UniProt: O31669
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulatory mechanism: S-box: transcription termination/ antitermination, the S-box riboswitch binds S-adenosylmethionine resulting in termination PubMed
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References